Abstract
Aim:
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a complex disease with different subtypes that affect patients’ quality of life. This study aim to evaluate the severity of CRSwNP and the risk of treatment resistance using the “Japanese epidemiological survey of refractory eosinophilic chronic rhinosinusitis” (JESREC) algorithm in outpatients at a university hospital.
Methods:
A retrospective study was conducted reviewing the medical records of CRSwNP outpatients. Clinical data including age, sex, blood eosinophilia, computered tomography (CT) scans, presence of asthma, and nonsteroidal anti-inflammatory drug (NSAID) use were assessed.
Results:
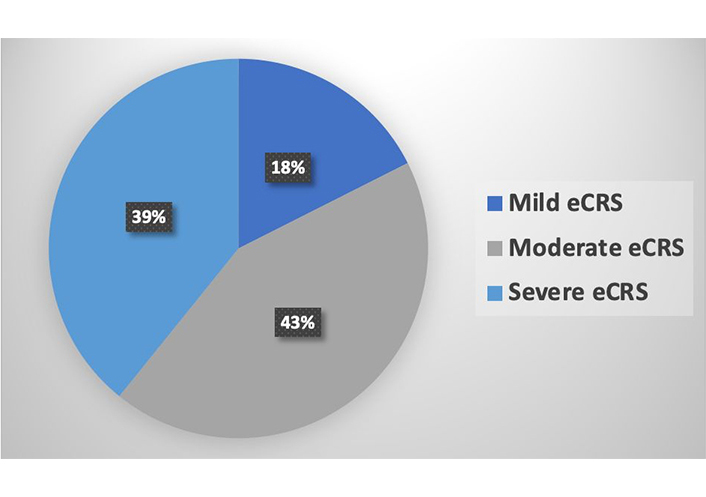
Medical records of 83 patients diagnosed with CRSwNP were analyzed, with 44 (53%) females and 39 (47%) males. The mean age was 61.8 years ± 14.1 years (range: 19–90 years). According to the JESREC algorithm, 9 (10.8%) patients were categorized as non-eosinophilic chronic rhinosinusitis (neCRS), and 74 (89%) were classified as eosinophilic chronic rhinosinusitis (eCRS). Among the eCRS patients, 13 (17.6%) were mild, 32 (43.2%) were moderate, and 29 (39.2%) were severe. Asthma was identified as a comorbidity in 57 patients (68.6%).
Conclusions:
A predominance of eCRS with moderate to severe risk of treatment resistance was confirmed. Considering the heterogeneity of chronic rhinosinusitis (CRS), the JESREC algorithm comes up as an instrument that uses objective criteria to assess higher risks of recurrency and refractoriness among patients before surgical treatment, helping to predict type2-driven biologics need.
Keywords
Chronic rhinosinusitis, nasal polyps, biomarkers, eosinophilic chronic rhinosinusitisIntroduction
Chronic rhinosinusitis (CRS) is an inflammatory disease of the nose and the paranasal sinuses defined in adults by the presence of nasal obstruction, post or anterior nasal drip, facial pain, and/or reduction or loss of smell for more than 12 weeks, associated with objective findings in nasal endoscopy and/or computered tomography (CT) [1–3]. It is generally classified according to the absence or presence of nasal polyps as CRS without nasal polyps (CRSsNP) and CRS with nasal polyps (CRSwNP) [4].
European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS) 2020 classify primary CRS as localized or diffuse, and each of them by endotype predomination as type 2 or non-type 2 and phenotypes [2], since CRS complexity is better represented by the endotypes and their variety of biomarkers that determine prognosis and treatments options [5]. The phenotypes reflect different cellular and molecular mechanisms, and for primary diffuse CRS, they are mainly eosinophilic CRS (eCRS) and non-eCRS (neCRS), distinguish by histologic eosinophilia with different cut points among countries [2, 4]. The mucosal eosinophilia brings some information about severity and response to therapies so many authors defend its use for clinic classify CRSwNP [4].
CRSwNP affects 2–4% of the general population and has a great impact on quality of life, as the burden of symptoms, associated diseases, long-term medical therapies, and need for surgery, negatively changes the physical, emotional, and social aspects of a patient’s daily life [6]. It is the most severe subtype of CRS with difficult management as recurrences are common even with the right clinical and surgical treatment, placing high costs on the public health system [6, 7].
The diagnosis of CRS relies on clinical evaluation, nasal endoscopy, and CT imaging findings. However, the differentiation between allergic rhinitis (AR) and non-AR (NAR) is crucial. Due to similar symptoms in both rhinitis types, diagnostic tools are necessary for accurate differentiation. CT is considered the gold standard imaging modality to identify the characteristic inflammatory pattern in CRS. Moreover, the Lund-Mackay score (LMS), with a maximum score of 24 or 12 per side, is used to grade the severity of the disease. An LMS score of 2 or less exhibits an excellent negative predictive value, whereas an LMS score of 5 or more shows an excellent positive predictive value, indicating the true presence of the disease. Nasal endoscopy is an essential diagnostic tool for CRS, providing high diagnostic accuracy [2].
The Japanese epidemiological survey of refractory eCRS (JESREC) was a multicenter retrospective study in Japan that evaluated what characteristics of CRS were related to recurrence or refractoriness, especially eosinophilic infiltration, and created an algorithm that allows CRS classification before any invasive treatment as biopsy or endoscopic sinus surgery (ESS) [4]. Assessing patient data and CT scan results they created a score that classifies CRSwNP as eosinophilic by a cutoff point ≥ 11, considering bilateral (diffuse) disease, presence of nasal polyps, blood eosinophilia, the dominant shadow of ethmoid sinuses in CT [4]. An algorithm has been developed to classify eCRS by the risk of refractoriness, considering blood eosinophilia > 5% and dominant shadow in ethmoid sinuses in CT and comorbidities: asthma and nonsteroidal anti-inflammatory drugs (NSAIDs) intolerance [4]. eCRS was classified into mild, moderate, and severe according to refractoriness risk, using only clinical data and imaging tests, helping establish a prognosis before treatment and the possibility of biological treatment need afterwards [4].
The significance of this study is in its potential to confirm and revolutionize the management of patients with CRS by employing a classification algorithm. The algorithm has the potential to decrease the number of invasive procedures required and aid in predicting the prognosis and potential need for biological treatments based on clinical evaluation, laboratory testing, and imaging studies. Consequently, we aim to utilize the JESREC algorithm to assess CRSwNP severity and risk of refractoriness among outpatients at a university hospital.
Materials and methods
In this study, we carried out a retrospective analysis of the medical records of CRSwNP outpatients previously diagnosed by nasal endoscopy, submitted to endoscopic nasal polypectomy, and received follow-up care from both immunology and ear, nose, and throat (ENT) departments at a university hospital. We collected clinical data including age, sex, and comorbidities such as asthma and/or intolerance to NSAIDs. Previous laboratory results were reviewed to assess for blood eosinophilia, while CT scans were examined for the presence of bilateral disease, nasal polyps, and the dominant shadow in the ethmoid sinuses using Lund-Mackay criteria, as in the original JESREC study. Patients without previous laboratory results or imaging tests in their hospital records were excluded from our analysis.
Results
We analyzed 150 CRSwNP outpatients followed up in immunology and ENT departments, and after exclusion 83 patients remained (55%). Forty-four (53%) were female and 39 (47%) were males. The mean age was 61.8 years ± 14.1 years (range: 19–90 years). Fifty-seven (68.6%) patients presented asthma as a comorbidity (Table 1).
Sample characteristics
| Characteristics | eCRS | neCRS | Total | |
|---|---|---|---|---|
| Sex | Male | 34 | 5 | 39 |
| Female | 40 | 4 | 44 | |
| Age | 61.8 ± 13.9 | 62 ± 16.26 | 61.8 ± 14.1 | |
| Asthma | 53 | 4 | 57 | |
| Mean blood eosinophils percentage | 6% | 3% | 5% | |
| CT (ethmoid ≥ maxillary) | 73 | 1 | 74 | |
According to JESREC score criteria, 9 (10.8%) patients were defined as neCRS and 74 (89%) as eCRS. Among eCRS patients, 13 (17.6%) were classified as mild, 32 (43.2%) moderate, and 29 (39.2%) severe by the refractoriness risk algorithm (Figure 1).
CT dominant shadow of the ethmoid sinus was present in 74 patients (89%). The mean eosinophils percentage in peripheral blood was 6% for eCRS and 3% for neCRS patients (Table 1). Considering only moderate and severe eCRS patients present 7% of blood eosinophils.
Discussion
CRS classification adopted in the past into CRSwNP and CRSsNP does not reflect the many inflammatory profiles involved, bringing up the necessity of endotyping to correctly reflect the disease complexity and improve its management [7, 8]. Endotyping systems have been proposed but they often demand invasive procedures such as nasal biopsy or surgical approach limiting, their practical use [8]. Since invasive procedures may not be available for everyone and around 40–60% of CRSwNP patients have asthma, and nearly 50% might have severe asthma making invasive procedures at great risk. The JESREC score criteria and algorithm have the major advantage of not requiring any procedures, and allowing clinicians to use clinical data, blood eosinophilia, and CT scans to diagnose eCRS and to presume the higher risk of recurrence and refractoriness in moderate and severe eCRS patients [9]. By assessing our patients’ records in a retrospective way, we were able to score 83 CRS outpatients of immunology and ENT departments without using EES or nasal biopsies, not currently available at our center, presenting as a valuable tool in institutions with fewer resources. We selected in our database patients with CRSwNP confirmed by imaging tests (CT and nasal endoscopy).
eCRS was diagnosed by JESREC score criteria in 74 (89%) patients, 61 (82.4%) with moderate and severe eCRS, with a higher risk of recurrence and refractoriness. We found an eosinophilic and moderate or severe predomination possibly because our institution receives highly complex cases to attend. Before JESREC, criteria for eCRS were vague, being determined by histopathological examination showing eosinophil cell infiltration in nasal mucosa with the variable cut of point according to the country. Now it is determined by a JESREC score ≥ 11 and confirmed by the presence of ≥ 70 eosinophils per high power field (HPF) [10].
Nasal mucosa eosinophilia is predominant in Western white patients, being less common in Asian countries [7]. It is associated in the literature with extensive sinus disease, less improvement in general and disease-specific quality of life, and higher recurrence rates, even when EES are performed appropriately [7, 9]. Severe eosinophilic infiltration was related to lower rates of disease control [10]. Bronchial asthma is also a common comorbidity in patients with eCRS [10]. In our study, 53 (71.6%) patients with eCRS presented asthma, that is another isolated factor associated with nasal polyps’ recurrence [7]. Mendelsohn et al. [11] reported 45% of recurrence and 25% of revision surgery after five years in patients with this comorbidity. Long-term follow up and more incisive and targeted therapies might be needed in those cases.
The limitation of this study is the fact that it is a chart review, although from an expressive number of patients. We were either not able to access nasal polyps recurrence since some patients had been submitted to surgery in other hospitals.
The main treatment for CRS is based on intranasal or systemic steroids and nasal irrigation, and when the symptoms persist EES is the gold standard and is performed to restore mucociliary clearance and sinus drainage [9, 12]. Even when well performed and followed by intranasal steroid use, some patients’ nasal polyps recur, showing that personalized treatment in CRS based on endotypes can improve assistance [9]. eCRS has a better response to steroids and biologics targeting type 2 cytokines, and its use can be considered [9].
Considering the heterogeneity of CRS, the JESREC algorithm comes up as an instrument that uses objective criteria to assess higher risks of recurrency and refractoriness among patients before surgical treatment, helping to predict type2- driven biologics needs. Thus, in the future, we intend to continue monitoring these patients and correlate with the actual need to use biologicals.
Abbreviations
| CRS: |
chronic rhinosinusitis |
| CRSwNP: |
chronic rhinosinusitis with nasal polyps |
| CT: |
computered tomography |
| eCRS: |
eosinophilic chronic rhinosinusitis |
| ESS: |
endoscopic sinus surgery |
| ENT: |
ear, nose, and throat |
| JESREC: |
Japanese epidemiological survey of refractory eosinophilic chronic rhinosinusitis |
| LMS: |
Lund-Mackay score |
| neCRS: |
non-eosinophilic chronic rhinosinusitis |
| NSAID: |
nonsteroidal anti-inflammatory drug |
Declarations
Author contributions
SDDJ, BVdOM, and PNF: Data curation, Writing—original draft. FCdC, KCdMC, and ESO: Data curation. JEF and SORV: Writing—original draft.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This research has been submitted and approved by Comitê de Ética em Pesquisa do Hospital Universitário Clementino Fraga Filho (HUCFF-UFRJ), number 090/09. The study was conducted in accordance with the Declaration of Helsinki.
Consent to participate
Informed consent to participate in the study was obtained from all participants.
Consent to publication
Not applicable.
Availability of data and materials
All datasets for this study are included in the manuscript.
Funding
Not applicable.
Copyright
© The Author(s) 2023.