Editor's Picks
Open Access
Review
Innovative regenerative genetic and nano therapies for diabetes and metabolic disease
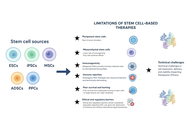
Diabetes and metabolic dysfunction represent growing global health challenges, and current therapies mitigate hyperglycemia without sufficiently altering the underlying disease processes. Rapid advances in regenerative medicine, gene editing, and nano-enabled drug delivery are reshaping therapeutic possibilities, offering the potential to restore β-cell function, enhance insulin sensitivity, and personalize care at an unprecedented scale. This review synthesizes emerging therapeutic strategies with the greatest translational promise, including stem cell-derived islet replacement, immune-evasive encapsulation devices, CRISPR-based gene correction, and targeted or glucose-responsive nanocarriers engineered for noninvasive insulin delivery. Complementary advances in multi-omics profiling, proteogenomics, microbiome science, and artificial intelligence are enabling stratification of patients based on molecular signatures, optimizing therapeutic selection, and predicting clinical outcomes. Despite remarkable breakthroughs—such as the early-phase clinical success of stem cell-derived β-cell replacement and organ-targeted lipid nanoparticles (LNPs)—significant barriers remain, including immunogenicity, off-target editing, scalability, ethical concerns, and long-term safety. By integrating mechanistic insights with translational developments, this review outlines a forward-looking perspective on next-generation treatments poised to shift diabetes care from glycemic management toward disease modification and personalized metabolic restoration.
Open Access
Original Article
Normal triglyceride levels are positively associated with plasma glucose levels and type 2 diabetes diagnosis in Chinese adults
Aim:
Hypertriglyceridemia is linked to increased risk of diabetes diagnosis, incidence, and mortality. However, whether individuals with normal triglyceride levels (i.e., < 1.7 mmol/L) uniformly exhibit low diabetes risk remains underexplored. Specifically, it is unclear whether triglyceride levels within the normal range are associated with plasma glucose levels and the prevalence of type 2 diabetes (T2DM). This study aimed to address these gaps by examining the associations between triglyceride levels and fasting plasma glucose, as well as between triglyceride levels and T2DM, in individuals with triglycerides in the normal range.
Methods:
This cross-sectional study included 16,706 Chinese adults with triglyceride levels below 1.7 mmol/L. Among them, 1,067 had T2DM. Associations between triglyceride levels and fasting plasma glucose were assessed using linear regression, while associations with T2DM were evaluated using binary logistic regression. The optimal triglyceride cut-off for T2DM diagnosis was determined via receiver operating characteristic (ROC) curve analysis.
Results:
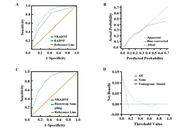
Triglyceride levels were positively associated with fasting plasma glucose after multivariate adjustment (β = 0.034, P < 0.001). A one-unit increase in the natural log of triglyceride levels was associated with a 61% higher adjusted odds of T2DM [odds ratio (OR), 1.61; 95% confidence interval (CI), 1.19–2.17; P = 0.002]. The optimal triglyceride cut-off for T2DM diagnosis was 1.09 mmol/L. Participants with triglyceride levels ≥ 1.09 mmol/L had a 28% higher odds of T2DM (OR, 1.28; 95% CI, 1.07–1.53; P = 0.006) compared to those with levels below the cut-off.
Conclusions:
Among individuals with normal triglyceride levels, higher triglyceride concentrations were associated with higher odds of T2DM diagnosis, with an optimal diagnostic cut-off of 1.09 mmol/L. These findings suggest that adults with triglyceride levels more than 1.09 mmol/L may benefit from closer monitoring for T2DM development.

Open Access
Commentary
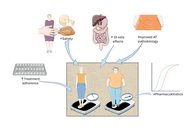
Treating obesity with GLP-1 RAs: does sex matter? A commentary on the meta-analysis by Yang et al. (J Diabetes 2025;17(3):e70063)
This commentary discusses a recent article (J Diabetes 2025;17(3):e70063), focusing on interpreting the study’s sex-stratified results in a broader clinical and mechanistic context. The authors of this systematic review and meta-analysis of 14 randomized trials demonstrate that women achieve greater weight loss induced by glucagon-like peptide-1 (GLP-1) receptor agonists compared to men (mean difference of 1.04 kg or 1.69%). Analyses specific to different drugs consistently show that women benefit more from dulaglutide, liraglutide, semaglutide, and retatrutide, with trials focused on obesity further emphasizing this gap. Sensitivity analyses confirm the reliability of these findings and indicate the absence of publication bias. We discuss the clinical implications of these results, suggesting that healthcare providers should consider sex differences when counseling, monitoring, and dosing patients. We also advocate for future trials that are adequately powered and stratified by sex to evaluate factors such as adherence, adverse events, and body composition. Mechanistic hypotheses, such as sex-related pharmacokinetics, estrogen-GLP-1 synergy, and varying inflammatory responses, should be investigated further to inform precision dosing. Lastly, we recommend that regulatory agencies revisit current labeling, which claims no sex differences, as more sex-stratified evidence becomes available. It is important to acknowledge the existing heterogeneity and remaining uncertainties in this area of research.
Articles
Latest
Most Viewed
Most Downloaded
Most Cited
Open Access
Review
Osteoarthritis from a female perspective
Maritza Vidal, Nancy E Lane
Published: April 09, 2026 Explor Endocr Metab Dis. 2026;3:101466

Open Access
Case Report
Epiploic appendagitis during rapid weight loss associated with tirzepatide therapy: a case report
Beshoy M. Astfanous, Jennifer D. Goldman
Published: April 02, 2026 Explor Endocr Metab Dis. 2026;3:101465
Open Access
Review
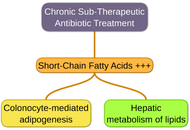
Obesity and antibiotic exposure: an overhyped theory
Valérie Lannoy
Published: March 18, 2026 Explor Endocr Metab Dis. 2026;3:101464
Open Access
Original Article
Development and validation of a novel risk assessment model for necrotizing fasciitis in patients with diabetic foot
Xun Li ... Qingfeng Cheng
Published: March 12, 2026 Explor Endocr Metab Dis. 2026;3:101463
Open Access
Original Article
Fat deposits and their relationship with type 2 diabetes in patients with metabolic syndrome
Ivonne G. Narváez-Ortiz ... Alberto Maceda-Serrano
Published: March 09, 2026 Explor Endocr Metab Dis. 2026;3:101462
This article belongs to the special issue Current Views on Pathogenesis, Diagnosis and Management of Type 2 Diabetes Mellitus and Its Complications and Related Conditions

Open Access
Review
Innovative regenerative genetic and nano therapies for diabetes and metabolic disease
Maria-Kalliopi Spanorriga ... Konstantinos Tsioufis
Published: March 08, 2026 Explor Endocr Metab Dis. 2026;3:101461
This article belongs to the special issue Innovative Strategies for Diabetes and Metabolic Disorders: Current and Future Directions

Open Access
Commentary
Updates from the 2025 American Diabetes Association guidelines on standards of medical care in diabetes
Dipti Tiwari ... Tar Choon Aw
Published: April 15, 2025 Explor Endocr Metab Dis. 2025;2:101428
Open Access
Commentary
The 2024 American Diabetes Association guidelines on Standards of Medical Care in Diabetes: key takeaways for laboratory
Dipti Tiwari, Tar Choon Aw
Published: July 23, 2024 Explor Endocr Metab Dis. 2024;1:158–166

Open Access
Review
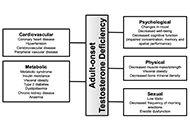
Adult-onset testosterone deficiency: the usefulness of hormone replacement in reducing mortality in men with this common age-related condition
Amar Mann ... Sudarshan Ramachandran
Published: June 28, 2024 Explor Endocr Metab Dis. 2024;1:83–100
This article belongs to the special issue The Fountain of Youth: Decoding the Hormonal Regulation of Aging
Open Access
Review
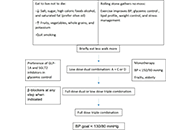
A brief approach to hypertension in type 2 diabetes mellitus
Yilmaz Gunes
Published: February 04, 2025 Explor Endocr Metab Dis. 2025;2:101422
This article belongs to the special issue Current Views on Pathogenesis, Diagnosis and Management of Type 2 Diabetes Mellitus and Its Complications and Related Conditions
Open Access
Review
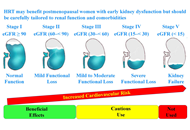
Optimizing hormone replacement therapy for postmenopausal women with type 2 diabetes: a review
Butheinah A. Al-Sharafi, Samih A. Odhaib
Published: April 28, 2025 Explor Endocr Metab Dis. 2025;2:101430
This article belongs to the special issue Metabolic Syndrome in Menopause
Open Access
Review
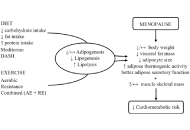
Healthy adipose tissue after menopause: contribution of balanced diet and physical exercise
Bruno Vecchiatto ... Fabiana S. Evangelista
Published: March 13, 2025 Explor Endocr Metab Dis. 2025;2:101424
This article belongs to the special issue Metabolic Syndrome in Menopause
Open Access
Review
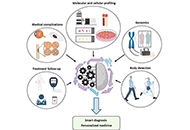
Recent advances in artificial intelligence-assisted endocrinology and diabetes
Ioannis T. Oikonomakos ... Stefan R. Bornstein
Published: April 01, 2024 Explor Endocr Metab Dis. 2024;1:16–26
Open Access
Commentary
Updates from the 2025 American Diabetes Association guidelines on standards of medical care in diabetes
Dipti Tiwari ... Tar Choon Aw
Published: April 15, 2025 Explor Endocr Metab Dis. 2025;2:101428

Open Access
Case Report
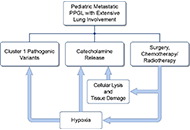
A case series of three patients with extensive lung metastatic pheochromocytoma/paraganglioma: evaluation, treatment challenges, and outcomes
Kailah M. Charles ... Karel Pacak
Published: November 15, 2024 Explor Endocr Metab Dis. 2024;1:218–233
Open Access
Case Report
Patient diagnosed with acromegaly and pituitary apoplexy after breast carcinoma treatment: challenges in diagnosis and management
Ignacio Jiménez Hernando, Laura González Fernández
Published: November 26, 2024 Explor Endocr Metab Dis. 2024;1:234–243
Open Access
Commentary
The 2024 American Diabetes Association guidelines on Standards of Medical Care in Diabetes: key takeaways for laboratory
Dipti Tiwari, Tar Choon Aw
Published: July 23, 2024 Explor Endocr Metab Dis. 2024;1:158–166

Open Access
Original Article
Comparative pharmacodynamic analysis of resmetirom, semaglutide and obeticholic acid in translational mouse models of MASH
Rufeng Zhang ... Chunli Sun
Published: June 24, 2025 Explor Endocr Metab Dis. 2025;2:101433
Open Access
Original Article
Waist-to-height ratio as a novel marker of metabolic syndrome in patients with type 2 diabetes mellitus
Elif Basaran, Gulali Aktas
Published: January 10, 2025 Explor Endocr Metab Dis. 2025;2:101421
This article belongs to the special issue Current Views on Pathogenesis, Diagnosis and Management of Type 2 Diabetes Mellitus and Its Complications and Related Conditions
Open Access
Commentary
Updates from the 2025 American Diabetes Association guidelines on standards of medical care in diabetes
Dipti Tiwari ... Tar Choon Aw
Published: April 15, 2025 Explor Endocr Metab Dis. 2025;2:101428

Open Access
Review
Synergistic glucocorticoids, vitamins, and microbiome strategies for gut protection in critical illness
Gianfranco Umberto Meduri
Published: May 14, 2025 Explor Endocr Metab Dis. 2025;2:101432
Open Access
Review
Recent advances in artificial intelligence-assisted endocrinology and diabetes
Ioannis T. Oikonomakos ... Stefan R. Bornstein
Published: April 01, 2024 Explor Endocr Metab Dis. 2024;1:16–26

Open Access
Commentary
The 2024 American Diabetes Association guidelines on Standards of Medical Care in Diabetes: key takeaways for laboratory
Dipti Tiwari, Tar Choon Aw
Published: July 23, 2024 Explor Endocr Metab Dis. 2024;1:158–166

Open Access
Review
Glucocorticoid receptor alpha: origins and functions of the master regulator of homeostatic corrections in health and critical illness
Gianfranco Umberto Meduri
Published: March 28, 2025 Explor Endocr Metab Dis. 2025;2:101426

Special Issues
Ongoing Special lssues
Completed Special lssues
Innovative Strategies for Diabetes and Metabolic Disorders: Current and Future Directions
Guest Editors: Dawood Khan; Victor Gault
Submission Deadline: May 20, 2026
Published Articles: 4

Current Views on Pathogenesis, Diagnosis and Management of Type 2 Diabetes Mellitus and Its Complications and Related Conditions
Guest Editor: Gulali Aktas
Submission Deadline: May 20, 2026
Published Articles: 8
Role of Dysregulated Cytokine Signaling Pathways in Metabolic Disease
Guest Editor: Alister C. Ward
Submission Deadline: May 27, 2026
Published Articles: 1
Oxidative Stress and Diabetes – Remedies through Functional Food
Guest Editor: Viduranga Y. Waisundara
Submission Deadline: May 27, 2026
Published Articles: 0

Metabolic Syndrome in Menopause
Guest Editor: Tzong-Shyuan Lee
Submission Deadline: May 27, 2026
Published Articles: 3
The Impact of Digitalization To Improve Nutrition and Self-Management in Patients With Diabetes
Guest Editor: Peter Schwarz
Submission Deadline: May 27, 2026
Published Articles: 1

The Fountain of Youth: Decoding the Hormonal Regulation of Aging
Guest Editor: Marijn Speeckaert
Submission Deadline: May 27, 2026
Published Articles: 5

Journal Information
Journal Indexing
Journal Metrics















 Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys
Title: Unravelling the interplaybetween #Harmattan wind andbaroreflex functions: implicationon environmental health andcardiovascular #pathophys


