Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
2Department of Gastrointestinal Surgery, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
ORCID: https://orcid.org/0000-0002-6364-0553
Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
Affiliation:
4Department of Clinical and Molecular Medicine, Sapienza University of Rome, Santo Andrea Hospital, Via di Grottarossa, 1035- 00189 Rome, Italy
Affiliation:
1Department of Medical Data Science, Center of Medical Innovation and Translational Research, Osaka University Graduate School of Medicine, Suita, Yamadaoka 2-2, Osaka 565-0871, Japan
Email: hishii@gesurg.med.osaka-u.ac.jp
ORCID: https://orcid.org/0000-0002-0632-6517
Explor Med. 2023;4:107–114 DOI: https://doi.org/10.37349/emed.2023.00127
Received: October 27, 2022 Accepted: November 21, 2022 Published: February 28, 2023
Academic Editor: Marco Ragusa, Università degli Studi di Catania, Italy
The article belongs to the special issue RNA World in Health and Disease
m6A RNA methylation, a predominant type of RNA modification, is involved in regulating mRNA splicing, stability, and translation as well as the interaction between nucleoproteins and noncoding RNAs. Recent studies have revealed that m6A RNA methylation plays a critical role in the self-to-non-self-recognition of immune cells against endogenous mutations in cancer and exogenous organism-related infections. As an epigenetic mechanism, m6A RNA modification induces immune cell signal transduction, which is altered in the tumor microenvironment, as detected in liquid biopsy. Furthermore, m6A RNA methylation-related inflammation is involved in the cellular response to viral infections, including the emerging severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) infection. Given the importance of the immune response in maintaining homeostasis in higher eukaryotes, m6A RNA methylation could be useful not only for the early detection of cancer but also for SARS-CoV-2 screening during a global pandemic.
Recently, RNA modification has emerged as a critical epigenetic mechanism involved in the regulation of embryonic stem cell development [1], immune cell response [2], and various conditions, such as infection [3] and cancer progression and development [4]. As a mechanism, RNA modification, the most common and critical in function and stability of which is N6-methylation, is reported to affect folding, stability, degradation, and multiple processes, such as splicing, peptide translation, export, and decay [5]. Furthermore, RNA modification is involved in cellular interactions [5]. The mechanism has been elucidated via the structural characterization of RNA methyltransferases (MTs) [5]. RNA methylation is characterized by three steps that are similar to those of histone modification, namely, writing, erasing, and reading. Thus, the dynamic and reversible chemical m6A RNA methylation may serve as a critical epigenetic marker of biological significance given the pivotal role of DNA and histone methylation in basic life processes [6]. MT-like 3 (METTL3; N6-adenosine-MT complex catalytic subunit) and METTL14 (N6-adenosine-MT subunit) form a stable heterodimeric core complex that functions in cellular m6A deposition on nuclear RNAs and catalyzes m6A RNA methylation [7]. Wilms’ tumor 1-associating protein (WTAP), a mammalian splicing factor, can interact with this complex and influence its methylation [7]. The core complex of METTL3–14 with WTAP induces methylation at N6-adenosines [8, 9]. Functional demethylases that reverse this methylation have been identified as erasers, including fat mass and obesity-associated (FTO) protein [10] and alpha-ketoglutarate-dependent dioxygenase AlkB homolog 5 (ALKBH5) [11]. However, several factors have been reported as readers, including heterogeneous nuclear ribonucleoprotein (hnRNP) [12] and YTH N6-methyladenosine RNA-binding proteins [YTH domain family (YTHDF)] [13]. Previous studies have indicated that m6A RNA methylation provides diagnostic and therapeutic targets for innovative medicine against human diseases such as cancer [14]. Nonetheless, recent studies have shown that m6A RNA methylation may also give rise to potential targets for the treatment of infectious diseases caused by severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) [15], a highly transmissible coronavirus that emerged in late 2019 and is now spreading globally [16]. This concept has several key implications. First, as a scientific implication, the biological response to exogenous and endogenous nucleic acids involves a shared mechanism of RNA methylation, which is one of the processes of biological homeostasis maintenance. Second, drugs developed for cancer treatment can be used to treat other diseases via repositioning, thereby significantly reducing the cost and time required for therapy development. Third, it is expected that if the mechanisms shared by multiple diseases can be used in diagnosis, social implementation in medical and health examinations could be accelerated.
The process of pathogen elimination by immunological activation includes rapidly sensing the invasion of pathogenic microorganisms. Hence, previous research on the sensing mechanism of biomaterials and many other signaling patterns has focused on toll-like receptors (TLRs), which has allowed the discovery of many pattern recognition receptors (PRRs) [17]. PRRs provide the structures of various constituent molecules derived from microorganisms, such as pathogen-associated molecular patterns, which consist of sugars, lipids, proteins, and nucleic acids [18]. In humans, PRRs have been reported to play a role in infections and also serve as sensors for internal abnormalities, such as biological processes of tissue damage and repair caused by stress or cancer [17]. Therefore, in response to dangerous conditions in an organism, the biological defense system is activated to quickly sense the hazard and emit a “danger signal,” thus maintaining homeostasis [19]. Previous research has shown that nucleic acid sensing mechanisms play critical roles in biological defense systems [20]. Compared with DNA recognition, the RNA recognition mechanism in the innate immune system is well characterized. Several RNA sensor molecules have been identified as their downstream signals, including cell membrane binding TLR3, TLR7, and TLR8 [21, 22] as well as cytoplasmic antiviral innate immune response receptors, such as retinoic acid-inducible gene I (RIG-I)’s product, interferon (IFN) induced with helicase C domain 1 such as melanoma differentiation-associated protein 5 (MDA5), and RIG-I-like receptor such as ATP-dependent helicase (LGP2) [23–26]. RIG-I is essential for RNA sensing, thus triggering antiviral and inflammatory responses that control RNA viral replication in response to virus-specific RNA structures [27]. Upon viral RNA recognition, RIG-I has been reported to recruit the adaptor protein, a mitochondrial antiviral signaling protein, on the outer mitochondrial membrane. This recruitment leads to a signaling cascade of type I IFNs [27]. RIG-I activation is tightly regulated via various post-translational modifications to prevent aberrant innate immune signaling. However, viruses have evolved evasion mechanisms, such as sequestering viral structures from RIG-I detections, which can target receptor or signaling molecules for degradation [27]. A previous study has demonstrated that RIG-I triggers an antiviral response upon binding non-self RNA with a stretch of double-stranded RNA bearing a base-paired 5’ppp nucleotide [28]. RIG-I adenosine triphosphatase activity can restrict the interaction between RIG-I signaling and non-self RNA [28]. Importantly, research on RNA modification has revealed that viruses acquire m6A in their RNA as a means of mimicking cellular RNA to avoid detection by innate immunity. This finding implies that the m6A modification enables viral RNA to evade RNA sensor RIG-I [29]. A study has suggested that viral m6A RNA modification can be used as a vaccine target to attenuate viral infections, such as those caused by human metapneumovirus [29]. Furthermore, a recent investigation has observed that METTL3 regulates viral m6A RNA modification and host cell innate immune responses during SARS-CoV-2 infection [30]. The study also reported that the SARS-CoV-2 genome contains m6A modifications enriched in the 3’ region of RNA and that RIG-I binds to SARS-CoV-2 viral RNAs and is inhibited by m6A RNA modifications [30]. In patients with severe coronavirus disease 2019 (COVID-19), METTL3 is reduced and inflammatory genes are induced [30], which agrees with the findings in other viruses [29]. It has been shown that host m6A components interact with viral proteins to modulate SARS-CoV-2 replication [31]. It has further been suggested that SARS-CoV-2 can cause an imbalance of methylated and unmethylated RNA via METTL3. This phenomenon may be involved in host recognition as well as in eliciting a severe immune response and the resultant adverse effects in patients with COVID-19. Further research is thus needed on the relationship between METTL3 activity and m6A modification status and biological response in patients with COVID-19.
The role of RNA methylation in cancer has been investigated [4]. Recent studies have revealed that RNA methylation is involved in the biological behaviors of glioblastoma stem cells [32], self-renewal of leukemic stem cells [33], therapeutic implications of coding and noncoding RNAs [34], and direct link between cancer metabolism and progression [35]. The m6A modification is associated not only with cancer but also with cardiovascular diseases [36] and Alzheimer’s disease [37]. Recent studies have revealed that small-molecule (STM2457) inhibition of METTL3 can serve as a favorable strategy against myeloid leukemia [38, 39]. Eltrombopag, a nonpeptide agonist of the active thrombopoietin receptor encoded by the myeloproliferative leukemia virus proto-oncogene (c-Mpl), is used to treat thrombocytopenia and has recently been reported as a selective allosteric inhibitor of the METTL3–14 complex [40].
The study of RNA methylation in microRNAs (miRNAs) and short noncoding RNAs revealed that RNA methylation at adenine and cytosine is relatively specific in gastrointestinal cancers, such as pancreatic cancer, as detected with mass spectrometry in serum via liquid biopsy and in surgically resected samples via solid biopsy [41]. Tunnel current sequencing analysis revealed that miRNAs derived from cancer cells had varying fingerprints in adenine and cytosine [42]. As exosomal miRNAs in liquid biopsy can be derived from tumor tissues as a whole-body response, it has been suggested that the profiling of RNA modification provides useful information not only for the early detection of cancer but also for determining the responses to chemotherapy, radiotherapy, and immunotherapy. Nonetheless, which methods are suitable for the diagnosis or monitoring of cancers in the pancreas [43], stomach [44], lung [45], colon, and rectum [46] is yet to be ascertained.
Recent studies have revealed that a volatile organic compound (VOC)–ozone mixture has a functional impact on RNA chemistry, including the prominent RNA oxidative modification 8-oxo-7,8-dihydroguanine, in lung epithelial cells. This finding offers a new insight into the relationship between atmospheric agents and cellular responses involved in human diseases [47], as being more consistent markers of cellular stress relative to other inflammation markers, such as interleukin 8 and lactate dehydrogenase [48]. In this regard, attempts have recently been made to utilize VOC-based methods for cancer diagnosis [49, 50]. Although several odorants have been detected with mass spectrometry, animal-based detection methods are capable of detecting multiple or complex signaling responses. Thus, such methods have the potential to pave the way for new modalities [51]. Although previous studies have demonstrated the efficacy of a low-cost cancer screening test using cancer-sniffing canines, a more affordable and accessible option is desired, especially for low-resourced populations [52]. A recent study found that a scent test using nematodes is effective in screening for early-stage pancreatic cancer [53]. These results indicate that the methods should be studied further for the detection of refractory cancers.
It has been shown that smell dysfunction to some extent is associated with COVID-19 in > 90% of the cases [54], a recent study found that non-neuronal expression of SARS-CoV-2 entry genes in the olfactory system is likely to be involved in the mechanisms underlying COVID-19-associated anosmia [55]. The exact mechanism in infected cells, i.e., viral RNA-mediated activation of PRRs or involvement of RIG-1 and MDA5, remains unknown (Figure 1).
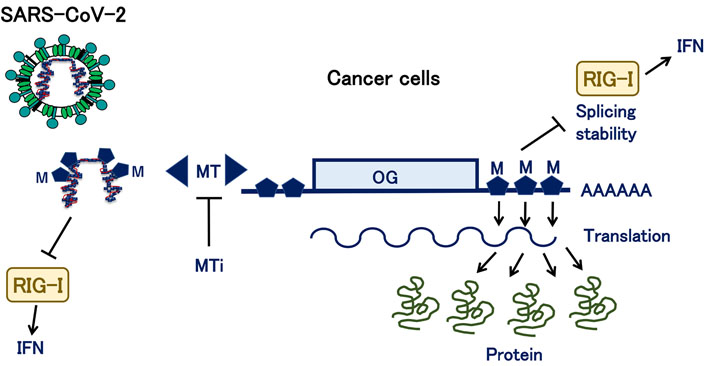
A schematic representation of the parallel involvement of RNA MT in cancer and COVID-19. OG: oncogene; MTi: MT inhibitors; M: methylated residue; A: multiple adenosine monophosphates in poly (A) tail
Although RNA modifications include many changes to the chemical composition of RNA molecules post-synthesis that have the potential to alter function or stability, previous studies indicated that methylation of RNA affects heterogeneity of malignant tumors [4]. Among the various RNA methylations, m6A RNA modifications has been characterized well as therapeutic targets [4]. Here we noted that a repositioning of m6A RNA targeting from cancers to infectious diseases may be possible. At first glance, cancer and COVID-19 appear unrelated, but they are entwined like a rope. First, as the common mechanism, RNA modification, which is important in cancer, plays a key role in the recognition of SARS-CoV-2 by the host cells and in the biological response to the infection. Second, epoch-making diagnostic and drug discovery technologies targeting RNA modifications, especially RNA N6-methylation, the most prevalent type of RNA methylation are expected to be used in the treatment of inflammation-based cancers and emerging infectious diseases.
Third, beyond the impact of COVID-19, the changes in people’s behavioral patterns globally cannot be ignored [56]. The global COVID-19 pandemic has not only had an imminent impact on routine medical care, including surgery, radiotherapy, and chemotherapy, but has also altered the cancer screening rates, thereby leading to poor early-stage cancer detection [57–59]. The pandemic eventually resulted in a sustained decrease in the number of people referred, diagnosed, and treated for cancer [59]. However, the impact on future cancer morbidity because of a failure of disease detection has been debated, and an analysis of long-term prognosis is awaited. The pandemic had a negative impact on cancer screening in primary care practice, but some were able to mitigate the effects by shifting their focus to processes that do not involve in-person office visits [57]. It is suggested that public health efforts are required to address the huge cancer screening deficit caused by the COVID-19 pandemic, including the increased use of noninvasive screening modalities [58].
METTL3, an RNA MT, is involved in the methylation of mRNA from OGs, which helps in regulating mRNA splicing, stability, and translation. The process is involved in the modulation of the cytoplasmic antiviral innate immune response receptors, such as RIG-I, which stimulates IFN response. In addition, as mentioned in the text, recent studies have shown that MT can methylate viral RNA from SARS-CoV-2, which is involved in the RIG-I-IFN pathway. MTi can antagonize the activity of MT, such as METTL3.
COVID-19: coronavirus disease 2019
IFN: interferon
METTL3: methyltransferase-like 3
miRNAs: microRNAs
MT: methyltransferase
PRRs: pattern recognition receptors
RIG-I: retinoic acid-inducible gene I
SARS-CoV-2: severe acute respiratory syndrome-coronavirus-2
TLRs: toll-like receptors
HS: Data curation, Writing—original draft, Writing—review & editing. T Hara: Data curation, Writing—original draft, Conceptualization, Methodology, Writing—review & editing. CO: Data curation, Writing—original draft, Writing—review & editing. YA: Data curation, Writing—original draft, Writing—review & editing. YT: Data curation, Writing—original draft, Writing—review & editing. YH: Data curation, Writing—original draft, Writing—review & editing. MO: Data curation, Writing—original draft, Writing—review & editing. EdL: Writing—original draft, Methodology, Writing—review & editing. T Hirotsu: Writing—original draft, Methodology, Writing—review & editing. AV: Writing—original draft, Methodology, Writing—review & editing. HI: Conceptualization, Data curation, Methodology, Writing—original draft, Writing—review & editing.
Partial institutional endowments were received from Taiho Pharmaceutical Co., Ltd. (Tokyo, Japan); Hirotsu Bio Science Inc. (Tokyo, Japan); Kinshu-kai Medical Corporation (Osaka, Japan); Kyowa-kai Medical Corporation (Osaka, Japan); IDEA Consultants Inc. (Tokyo, Japan); and Unitech Co. Ltd. (Chiba, Japan). EdL is an employee and T Hirotsu is the CEO of Hirotsu Bio Science Inc.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology [17cm0106414h0002; JP21lm0203007; 18KK0251; 19K22658; 20H00541; 21K19526; 22H03146; 22K19559; 16H06279 (PAGS)]. Partial support was offered by Mitsubishi Foundation to HI. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2023.
Copyright: © The Author(s) 2023. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Cristina Barbagallo ... Marco Ragusa
Kenneth Lundstrom
Azizul Haque, Anudeep B. Pant
