Abstract
Aim:
Childhood obesity is accompanied by an increased prevalence of abnormal glucose tolerance (AGT) including the prediabetes states. This study aims to investigate and evaluate the use of oral glucose tolerance test (OGTT) for detecting AGT among overweight and obese children.
Methods:
A retrospective study was conducted on 895 overweight and obese Chinese children (6–18 years) with obesity assessment and analysis of demographic, anthropometric, and biochemical parameters data between January 2006 and December 2015 at Tseung Kwan O Hospital, Hong Kong Special Administrative Region.
Results:
The proportion of males and older age group was 63.7% and 55.9%, respectively. Girls were more in older age groups (62.7% vs. 52.0%, P = 0.002). AGT occurred in 17.1% of the cohort [impaired glucose tolerance (IGT) was the most frequent morbidity (11.3%)]. After regression analysis, female sex, low-density lipoprotein (LDL), triglyceride (TG), older age group, and homeostasis model assessment of insulin resistance (HOMA-IR) ≥ 4.1 were significantly associated with AGT.
Conclusions:
AGT is common in overweight and obese Chinese children. Girls, older age, higher LDL, TG and HOMA-IR ≥ 4.1 showed significant association with AGT. OGTT is essential and fit-for-purpose to detect AGT in overweight and obese children.
Keywords
Pediatric obesity, abnormal glucose tolerance, diabetes mellitus, prediabetes, oral glucose tolerance testIntroduction
The prevalence of obesity in children and adolescents has increased thus becoming an epidemic in developed countries that has led to a worldwide increase in the rate of abnormal glucose tolerance (AGT) in this age group [1]. According to the statistics from the Health Service of the Department of Health, Hong Kong, the prevalence of childhood obesity amongst students rose from 15% in school year 1996/97 to 18.4% in 2016/17 [2]. Most patients with type 2 diabetes mellitus (T2DM) are obese and the onset is thought to occur subsequent to glucose metabolism alterations or prediabetes states, including impaired fasting glucose (IFG) and impaired glucose tolerance (IGT) [3]. This process may take decades to develop in adults but appears to be accelerated in children and adolescents. In a longitudinal study from the USA, 24% of obese children with IGT progressed to T2DM within 2 years [4]. Oral glucose tolerance test (OGTT) is well known as the gold standard for diagnosing glucose intolerance and diabetes in adult [5]. However, its use in children remains inconsistent, variable, and controversial [6]. The aim of our study was to investigate and evaluate the use of OGTT for detecting AGT among overweight and obese children. By identifying the prevalence, characteristics, and possibly risk factors, it may provide opportunities for early interventions and the subsequent prevention of the risk of developing diabetes and cardiovascular diseases in the future.
Materials and methods
This was a retrospective study of 895 overweight and obese Chinese children and adolescents aged between 6 and 18 years who had undergone obesity assessment between January 2006 and December 2015 in a regional hospital of Hong Kong. They were mainly referred from general practitioners, paediatricians, and governmental student health service for the assessment and management of obesity. A multidisciplinary approach involving dieticians and physiotherapists was adopted and they were assessed according to our local protocol.
Data obtained during routine clinical care were collected and analyzed anonymously. The detailed methodology for anthropometric measurement was described in our previous study [7]. Briefly, each patient underwent a physical examination with anthropometric measurements and acanthosis nigricans (AN) documented. Exclusion criteria included obesity related to known medical causes and patients taking medications that may alter metabolic profiles. Body mass index (BMI) was computed from body weight (kg) divided by the square of height (m2). The BMI percentiles of 90th and 97th centiles were considered overweight and obesity respectively. The percentile standards and calculation of z-score were based on a local survey in 1993. Blood pressure (BP) was measured in daytime with the children seated and rested. Two measurements were averaged for analysis. Z-score were calculated using local reference data [7].
All patients had overnight fast for at least 8 h while fasting plasma glucose (PG), insulin, lipid profile, and alanine aminotransferase (ALT) levels were obtained on the following day from 8 a.m. to 9 a.m., followed by a standard OGTT with 1.75 g glucose per kg body weight (up to a maximum 75 g was given). A second blood sample for PG was drawn 120 min afterwards (2-h PG). The homeostasis model assessment of insulin resistance (HOMA-IR) was calculated as the product of fasting serum insulin (μU/mL) and fasting PG (mmol/L) divided by 22.5 [8].
AGT was defined according to the American Diabetes Association (ADA) [3]. These include IFG (which refers to the fasting PG value of 5.6 mmol/L to 6.9 mmol/L), IGT (which refers to a 2-h PG value of 7.8 mmol/L to 11.0 mmol/L) and T2DM (diagnosed when the fasting PG value was equal to or higher than 7.0 mmol/L or 2-h PG value of 11.1 mmol/L or higher).
This study was reviewed and approved by the Hospital Authority Research Ethics Committee (Kowloon Central/Kowloon East, Reference No.: KC/KE-19-0043/ER-4).
Statistical analyses
All analyses were performed using IBM SPSS Statistics for Macintosh, Version 26 (IBM Corporation, Armonk, New York, USA) and statistical significance was accepted at P < 0.05 level. Descriptive analyses were performed using means and standard deviations (SDs) for continuous variables and frequencies with percentages for categorical variables. Student’s t-test was used for the between-groups comparison of means, for parametric variables while chi-square test was used to analyze the categorical variables. Logistic regression was used to estimate the odds ratio (OR) of having AGT. Variables included in the final adjusted regression model were those found at univariate analyses at P < 0.1 while closely interrelated variables including glucose and HOMA-IR were excluded. Goodness-of-fit of the regression model was assessed with the Hosmer-Lemeshow test. By using receiver operating characteristic (ROC) analyses, the optimal HOMA-IR (that is the maximum vertical distance between the ROC curve and the reference or chance line [maximum (sensitivity + specificity–1)]) was obtained.
Results
The overall characteristics of the 895 children were shown in Table 1. The mean age was 12.3 years, 571 (63.7%) were males, 500 (55.9%) children were in the older age group (12–18 years), 752 (84.0%) were obese, and 495 (55.3%) had AN. There were more females in the older age group than males.
Overall characteristics of the 895 children
| Characteristics | Boys (n = 571) | Girls (n = 324) | Total (n = 895) | P-value |
|---|---|---|---|---|
| Age (years) | 12.1 ± 2.8 | 12.6 ± 2.8 | 12.3 ± 2.8 | P = 0.012 |
| Age group (%) | P = 0.002 | |||
| 6–11 years | 48.0 | 37.3 | 44.1 | |
| 12–18 years | 52.0 | 62.7 | 55.9 | |
| BMI (%) | P = 0.096 | |||
| Overweight 90–97% | 17.5 | 13.3 | 16.0 | |
| Obese > 97% | 82.5 | 86.7 | 84.0 | |
| AN (%) | 55.7 | 54.6 | 55.3 | P = 0.943 |
a Student’s t-test; b chi-square test; c AN status was available in 884 children (98.8%); * statistically significant with P < 0.05
AGT occurred among 150 (17.1%) children, and IFG, IGT, and asymptomatic T2DM were 4.1%, 11.3%, and 1.7%, respectively (Table 2). Among those with AGT, 11 had both IFG and IGT, T2DM was diagnosed using the fasting glucose criteria in 4 children, while the remaining 11 children were identified using the 2-h glucose level. Females had a higher proportion of AGT status than males, with IGT contributing the highest.
Glucose tolerance status
| Glucose tolerance status | Boys (n = 562) | Girls (n = 317) | Total (n = 879) | P-value |
|---|---|---|---|---|
| AGT (%) | 13.7 | 23.0 | 17.1 | P < 0.001 |
| IFG | 3.6 | 5.0 | 4.1 | P = 0.285 |
| IGT | 8.7 | 15.8 | 11.3 | P = 0.001 |
| DM | 1.4 | 2.2 | 1.7 | P = 0.388 |
Glucose tolerance status was available in 879 children (98.2%). Eleven children had both IFG and IGT and these children were included in the IFG group. φ IFG includes IFG alone or IFG with IGT; ‡ DM was identified in 4 patients using the fasting glucose criteria while the remaining 11 patients were identified using the 2-h glucose in the OGTT; a chi-square test; * statistically significant with P < 0.05
The baseline characteristics and biochemical parameters were compared between the abnormal and normal glucose tolerance groups (Table 3). Subjects with AGT were significantly older, with higher prevalence among the females, had more AN, heavier, taller, higher BMI, as well as higher mean systolic and diastolic BP. However, BMI z-score, systolic BP z-score, and diastolic BP z-score did not differ between the two groups. As expected, the AGT group had higher fasting and 2-h post-OGTT glucose levels, higher fasting insulin, HOMA-IR, ALT, total cholesterol, low-density lipoprotein (LDL), triglyceride (TG), and lower high-density lipoprotein (HDL) levels.
Comparisons between the abnormal and normal glucose tolerance groups
| Variables | AGT (n = 150) | Normal glucose tolerance (n = 729) | P-value |
|---|---|---|---|
| Age (years) | 13.3 ± 2.6 | 12.1 ± 2.8 | < 0.001 |
| Age group (%) | 0.001 | ||
| 6–11 years | 32.0 | 46.5 | |
| 12–18 years | 68.0 | 53.5 | |
| Sex | < 0.001 | ||
| Male | 13.7 | 86.3 | |
| Female | 23.0 | 77.0 | |
| AN | 68.7 | 53.4 | < 0.001 |
| Body weight (kg) | 72.7 ± 16.6 | 66.6 ± 18.7 | < 0.001 |
| Body height (cm) | 156.8 ± 10.6 | 152.5 ± 14.5 | < 0.001 |
| BMI (kg/m2) | 29.2 ± 4.3 | 28.0 ± 3.9 | < 0.001 |
| z-score | 2.3 ± 0.4 | 2.2 ± 0.4 | 0.790 |
| Mean systolic BP (mmHg) | 119.1 ± 10.4 | 115.8 ± 9.6 | < 0.001 |
| z-score | 0.9 ± 0.9 | 0.8 ± 0.9 | 0.460 |
| Mean diastolic BP (mmHg) | 63.6 ± 7.5 | 61.2 ± 7.2 | < 0.001 |
| z-score | –0.2 ± 0.8 | –0.3 ± 0.9 | 0.072 |
| Fasting glucose (mmol/L) | 5.3 ± 0.7 | 4.9 ± 0.3 | < 0.001 |
| Postprandial glucose (mmol/L) | 8.7 ± 1.7 | 6.0 ± 0.9 | < 0.001 |
| Fasting insulin (μU/mL) | 26.1 ± 15.8 | 17.1 ± 11.4 | < 0.001 |
| HOMA-IR | 6.3 ± 4.6 | 3.7 ± 2.6 | < 0.001 |
| HOMA-IR ≥ 4.1 (%) | 65.7 | 32.5 | < 0.001 |
| ALT (units/L) | 44.0 ± 56.5 | 32.3 ± 37.7 | 0.002 |
| Total cholesterol (mmol/L) | 4.8 ± 0.9 | 4.6 ± 0.8 | 0.003 |
| HDL (mmol/L) | 1.2 ± 0.3 | 1.3 ± 0.4 | 0.010 |
| LDL (mmol/L) | 2.8 ± 0.8 | 2.6 ± 0.8 | 0.005 |
| TG (mmol/L) | 1.6 ± 0.7 | 1.4 ± 0.7 | < 0.001 |
∞ Percentage of glucose tolerance status within sex group; @ AN status was available for analysis in 869 children; a chi-square test, expressed in median (IQR); b student’s t-test, expressed in mean ± SD; * statistically significant with P < 0.05. IQR: interquartile range
Findings of further analysis of the risk factors of AGT using the stepwise multiple regression model are shown in Table 4. Female sex [OR = 1.888; 95% confidence interval (CI): 1.281–2.784; P = 0.001], LDL (1.484; 1.170–1.882; P = 0.001), TG (1.437; 1.074–1.924; P = 0.015), older age group (1.562; 1.044–2.338; P = 0.030), and HOMA-IR ≥ 4.1 (3.374; 2.261–5.034; P < 0.001) were significant independent variables associated with AGT.
Associations between clinical and laboratory parameters and AGT by multiple regression analyses
| Parameter | P-value | OR (95% CI) |
|---|---|---|
| Female sex | 0.001 | 1.888 (1.281–2.784) |
| LDL | 0.001 | 1.484 (1.170–1.882) |
| TG | 0.015 | 1.437 (1.074–1.924) |
| Age group (12–18) | 0.030 | 1.562 (1.044–2.338) |
| HOMA-IR ≥ 4.1 | < 0.001 | 3.374 (2.261–5.034) |
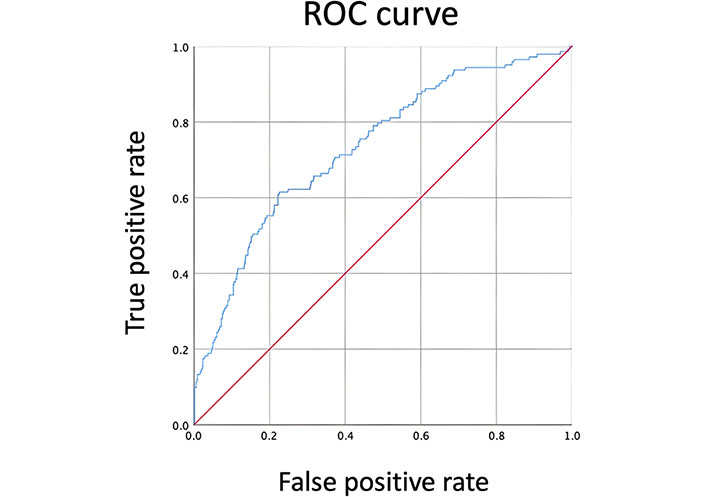
Using ROC analysis (Figure 1), the optimal HOMA-IR was 4.1, and the area under the curve was 0.734, with the sensitivity of 66% and specificity of 68%.
Discussion
Childhood obesity is one of the most serious public health challenges of the 21st century. The problem is global and the prevalence has increased at an alarming rate. A recent report published before the coronavirus disease 2019 (COVID-19) pandemic estimated that 158 million children are living with obesity in 2020 and the number will rise to 254 million in 2030 [9]. This would be further exacerbated by the COVID-19 pandemic response and a more drastic rise in childhood obesity will be imminent [10]. Driven by overweight and obesity, the prevalence of T2DM has also been increasing globally [1]. Diabetes is a well-recognized cause of premature mortality and morbidity, and it increases the risk of cardiovascular disease, kidney failure, blindness, and lower-limb amputation [11]. It frequently goes undiagnosed for many years without any classical diabetes symptoms. However, those undiagnosed patients might already have had increased risk of developing diabetes complications. T2DM in these patients seems to occur following the progression of the prediabetes states, and early detection of these obese children with prediabetes can ensure diabetes prevention and the prevention of other morbidities [3, 12].
In the present study, AGT is very common and is found in 17.1% of the cohort using OGTT. Female sex, older age, higher LDL, TG and HOMA-IR ≥ 4.1 have significant association with AGT in regression analysis.
It is important to note there is substantial controversy and variability regarding approaches to evaluate IR both in the literature and in clinical practice [6, 13, 14]. In general, HOMA-IR is the most commonly used surrogate marker in clinical practice and clinical research [6, 15]. Despite its popularity, the cutoff points for diagnosis of IR have not been defined yet in children [16]. Similar to HOMA-IR, Matsuda insulin sensitivity index also has strong correlations with the hyperinsulinemic-euglycemic clamp [17]. It is a more elaborate measurement derived from the OGTT and the correlation is more robust [18]. However, it requires more blood sampling so as to obtain the mean OGTT glucose and mean OGTT insulin concentration. To complicate the discussion further, there is also no universally accepted use of OGTT for detecting AGT in children and adolescents [6]. This is where our study seeks to provide insight.
AGT, as suggested, is caused by IR, defined as dysfunctional cellular response to insulin combined with pancreatic β-cell insufficiency and these also underlie the metabolic syndrome, which refers to a cluster of obesity-associated conditions including dyslipidaemia, fatty liver, hypertension, and cardiovascular disease [19]. The prevalence of AGT is highly variable in different studies and countries. Its prevalence was 17.1% in our cohort. In a study performed in Italy, the frequency of AGT was 12.4% [20], while another study from USA showed a total of 25% of children and 21% of the adolescents had IGT though this study was limited by its small sample size [21]. Rosenbaum et al. [22] found racial/ethnic differences in fractional body fat content and insulin secretory capacity, whereas East Asian children had a higher percentage of body fat at any BMI and lower insulin sensitivity than Caucasians. In our cohort, females were more in older age group, and with a higher prevalence of AGT. These findings might be attributed to the pubertal growth spurt which started earlier in girls. The relatively larger female size might have evolved to support the birth of offspring in adulthood [23]. Their higher percentage body fat therefore influenced the tendency of having AGT.
It is known that AGT occurs more in the older age group because puberty would reduce the insulin sensitivity through an increase in growth hormone, testosterone, and estrogen secretions, resulting in more fat mass [24]. The association between high BP and AGT has been reported previously. High BP and obesity are individually associated with an increase in odds of AGT by 1.8 and 2.2 folds respectively and even their coexistence increases the risk of AGT further [25]. This association could be explained by hyperglycemia causing vascular change or renal damage leading to high BP [26].
AN, a cutaneous marker of IR, can offer a visual cue that can help in the early diagnosis of underlying cardiometabolic disease [27]. In our study, it is found more common among those with AGT. Elevated ALT may indicate non-alcoholic fatty liver disease (NAFLD) in obese patients and a study showed that a high-normal ALT level independently increased to nearly three-fold the risk of having NAFLD [28]. IR affects the endogenous glucose production, glucose disposal, lipolysis, and lipid oxidation, which further increases the hepatic free fatty acids supply and increases lipid oxidation. This mechanism will subsequently lead to free radical formation causing liver cell injury, inflammation, and fibrosis [29, 30]. IR also plays a role in lipid metabolism with an enhanced synthesis of the hepatic very low density lipoprotein which could explain the increased plasma TG and LDL levels in obese children [31].
IR or AGT is believed to be related to the increased lipids in organs. Youths with T2DM were found to have three times the amount of liver fat compared with BMI-matched non-diabetic controls [19]. It has been suggested that genetic factors exert a large influence on the location of storage of excess lipids which determines the future development of IR [19].
Using multivariate analysis, we demonstrated that female sex, LDL, TG, age, and HOMA-IR more than 4.1 are independent factors of AGT. We believe that it is of great benefit to our children and adolescents if a prediction model based on these factors can be developed to facilitate the screening and assessment of AGT among obese children.
Recently, studies have attempted to use HOMA-IR, glycated hemoglobin (HbA1c), and fasting glucose to replace the OGTT in the assessment of IR and AGT. HOMA-IR has been widely used in clinical research to assess IR, and its use has been validated in children and adolescents [7, 32]. It provides a more physiological estimate of glucose homeostasis and a more convenient way to quantify IR than the gold standard euglycemic clamp method [33]. Nevertheless, there is no worldwide consensus on the cutoff values of HOMA-IR for IR and its value differs by race, ages and gender [16]. Different studies used different cutoff values for HOMA-IR for IR: Singh et al. [34], Keskin et al. [32] used 2.5 and 3.16, respectively, while Ng et al. [7] and Nsiah-Kumi et al. [35] set the value at 4, taking into account the physiological IR during puberty. Using the ROC curve, we obtained a cutoff value of HOMA-IR at 4.1, providing a satisfactory sensitivity of 66% and specificity of 68% in diagnosing AGT among Chinese children and adolescents. Further studies are needed to verify this cutoff value of HOMA-IR for AGT.
HbA1c has also been proposed to diagnose diabetes and prediabetes in adults [3]. HbA1c is an indicator of the average blood glucose concentration over the past three months. The test has the following advantages: stable at room temperature, less day-to-day variations, no fasting is required, and, only a single blood drawn. However, ethnic or racial differences in HbA1c levels do exist and medical conditions affecting the red cell turnover, including anemia, hemoglobinopathies, post-blood transfusion, and malaria can also interfere with the validity [36–38].
In children, the use of HbA1c in the diagnosis of diabetes remains controversial [37]. The adult recommendation of HbA1c to diagnose diabetes is based on epidemiological studies from adult population [3]. Studies using adult cutoff values suggested an underestimation of the prevalence of diabetes and prediabetes [37, 39]. Recently, Khokhar et al. [40] found that HbA1c alone is a poor differentiator of prediabetes in a group of obese Caribbean and African-American children. Hence, it remains unclear if HbA1C and the same cutoff value should be used in children [3, 37, 38].
Fasting glucose alone has also been proposed for the screening of high-risk cases [41]. However, our findings confirmed that IGT is the most common abnormal glucose metabolism status among obese children; screening with fasting glucose alone would fail to identify a large portion of at risk children. Our data demonstrated that 73.3% of silent diabetes (11 out of 15) and 73.3% of prediabetes (110 out of 150) would be missed.
Various strategies were also proposed to triage and stratify children for OGTT. Van der Aa et al. [42] proposed using combined fasting glucose and HOMA-IR (≥ 3.4) to triage patients for OGTT. They concluded with no false-negatives for diabetes and only 36% of IGT was missed. Applying the same approach with HOMA-IR ≥ 4.1 in our study, we still failed to identify 33.3% of diabetes and 34.0% of prediabetes (data not shown). Similarly, Poon et al. [43] derived a clinical pathway to stratify risk of AGT in children using family history and non-fasting sample (HbA1c and ALT). They were able to reduce 171 OGTTs in their cohort of 332 children. However, 18.3% (11 cases) of children with AGT will be overlooked.
Over the past decades, the utilization of OGTT in children has long been argued [6, 44, 45]. OGTT is described as time and labor-intensive by some [21]. Contrary to popular belief, OGTT was well tolerated in our cohort (failure rate of 0.2% due to vomiting after glucose load). Until a well-validated screening test is found, we believe OGTT is still fit-for-purpose in children.
Last but not least, early identification of this group of children with AGT represents a golden opportunity for intervention and follow-up. Intensive lifestyle modifications using multidisciplinary team approach had shown initial promising results [46, 47]. These approaches focusing on dietary modification and physical activity should always be the cornerstone of management. Additionally, Metformin has been evaluated in clinical trials of children with prediabetes [14]. It is a biguanide derivate and is considered relatively safe and effective in clinical trials, although most of the trials were of duration less than 18 months with small sample sizes. When added to lifestyle modification, clinical trials have shown effects of further decreasing BMI [47, 48].
The main strength of this study is the large sample size of children from the same ethnicity. Also, the documentation of AN is essentially complete and we have OGTT done in all of our children. However, it has several limitations. Pubertal staging, waist circumference, waist-to-height ratio, and family history were not documented and hence comprehensive analysis could not be performed. In addition, the data are from a single center and causality cannot be inferred due to the cross-sectional nature of the study.
In conclusion, the results of our study reaffirm AGT is common in overweight or obese children. We also found female sex, older age, HOMA-IR larger than 4.1, LDL and TG are five important factors linked to the development of AGT in overweight and obese Chinese children and adolescents. We postulate HOMA-IR, HbA1c, fasting glucose, and family history either alone or in combination are inadequate to assess children for prediabetes and T2DM. An OGTT is crucial and fit-for-purpose in identifying AGT among obese children. This study can contribute to the discussion on the proper use of OGTT and help paediatricians investigate appropriately in children with obesity.
Abbreviations
| AGT: | abnormal glucose tolerance |
| ALT: | alanine aminotransferase |
| AN: | acanthosis nigricans |
| BMI: | body mass index |
| BP: | blood pressure |
| HbA1c: | glycated hemoglobin |
| HOMA-IR: | homeostasis model assessment of insulin resistance |
| IFG: | impaired fasting glucose |
| IGT: | impaired glucose tolerance |
| LDL: | low-density lipoprotein |
| OGTT: | oral glucose tolerance test |
| OR: | odds ratio |
| PG: | plasma glucose |
| ROC: | receiver operating characteristic |
| T2DM: | type 2 diabetes mellitus |
| TG: | triglyceride |
Declarations
Author contributions
VYHL, JHMY, HYN, and LTWC: Conceptualization, Investigation, Writing—original draft, Writing—review & editing. All authors read and approved the submitted version.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This study was reviewed and approved by the Research Ethics Committee (Kowloon Central/Kowloon East) of Hospital Authority, Hong Kong Special Administrative Region (Reference No.: KC/KE-19-0043/ER-4).
Consent to participate
The need for patient consent was waived by the Research Ethics Committee (Kowloon Central/Kowloon East) of Hospital Authority for this retrospective cohort study because no personal identifiers or related information were obtained during the data collection process.
Consent to publication
The need for patient consent was waived by the Research Ethics Committee (Kowloon Central/Kowloon East) of Hospital Authority for this retrospective cohort study because no personal identifiers or related information were obtained during the data collection process.
Availability of data and materials
All relevant data are contained within the manuscript.
Funding
Not applicable.
Copyright
© The Author(s) 2023.