Abstract
Aim:
Gasserian ganglion (GG) percutaneous neurostimulation is a key resource in treating chronic refractory facial pain. However, this procedure has complications, and the literature does not yet document a gold standard for tunneling techniques. The main objective of this study was to compare two different tunneling techniques for GG stimulation and report whether there were associations between the type and rate of complications.
Methods:
This was a retrospective analysis of medical records of all patients with refractory facial pain, who underwent GG stimulation between January 2010 and June 2022. From 2010 to 2015, the percutaneous GG stimulation procedure was performed with a tunneling technique which involved passing through the preauricular region. Since 2016, this procedure has been performed with a tunneling technique that involved passing directly into the patient’s neck.
Results:
GG stimulation for refractory facial pain was performed in 49 patients. One of them was implanted twice because of bilateral trigeminal neuropathy. The ear tunneling technique was used for 20 GG stimulation procedures while the tunneling technique involving direct passage of the lead into the neck was used for 30 procedures. The comparison of these two different tunneling techniques showed no statistically significant difference in terms of complications incurred (Fisher exact test, p = 0.451). In the group of patients with ear tunneling, erosion and fibrosis of the neck skin occurred in 20% of cases. This type of complication is not present in patients in whom the other tunneling technique was used.
Conclusions:
Both tunneling techniques were found to be safe, but the direct technique does not cause fibrosis and erosion of the neck tissues as a complication.
Keywords
Gasserian ganglion stimulation, trigeminal neuropathic pain, atypical facial pain, complications, tunnelingIntroduction
Facial pain syndromes can be very difficult to manage and often require a multidisciplinary approach [1]. Trigeminal pain, especially trigeminal neuralgia, is the most common form of facial pain [2]. Medications could lead to pain control in 90% of patients with essential trigeminal neuralgia but other types of trigeminal pain, such as trigeminal neuropathic pain and atypical facial pain, are less responsive to medical treatments [3]. Neuromodulation represents a significant opportunity for better management of chronic refractory trigeminal pain but, to date, there are no neuromodulation devices commercialized specifically for facial pain syndromes [4]. Curiously, the earliest recorded use of neurostimulation was for facial pain in 1962 by Shelden, who performed mandibular branch neuromodulation years before the publication of the gate theory and spinal cord stimulation [5]. Gasserian ganglion (GG) stimulation by percutaneous approach is less invasive than the open surgical approach by craniotomy, and it has been optimized by the use of computed tomography (CT) based electromagnetic neuron avigation guidance for easier cannulation of foramen ovale [6]. Given the complex anatomy of the GG and its proximity to delicate structures, anatomical landmarks play a crucial role in percutaneous procedures through the foramen ovale, ensuring both precision and safety [7, 8].
Although there are good results in terms of pain relief, the GG neuromodulation technique to reduce the type and frequency of complications remains unknown [9]. In addition, tunneling of the leads through the facial tissue to the implant site remains a challenge due to the aesthetic outcomes and post-implant pain in a region already afflicted by intractable pain [4].
The present study aimed to compare two different tunneling techniques for GG stimulation and report whether there were associations between the type and rate of complications.
Materials and methods
The local Ethical Committee of Vitaz (Sint-Niklaas, Belgium) approved the study (EC22022). An informed consent form was not required due to the retrospective observational nature of the study. This was a retrospective analysis of medical records of all patients with refractory facial pain, who underwent GG stimulation at Multidisciplinary Pain Center of Vitaz between January 2010 and June 2022. A retrospective chart review was performed to obtain demographic information, facial pain history, and symptoms, imaging, history of previous surgeries, time and technique of GG stimulation, outcomes, and complications. All patients older than eighteen years old who underwent GG neuromodulation during the study period were considered.
Study population
GG stimulation for refractory facial pain was performed in 49 patients. One of them was implanted twice because of bilateral trigeminal neuropathy. All patients suffered from orofacial pain attributed to a lesion or disease of the trigeminal nerve, according to the new ICOP classification [10]. None of them suffered from classical trigeminal neuralgia and 11 patients suffered from trigeminal neuralgia with concomitant continuous pain. According to Burchiel’s classification, all patients suffered from trigeminal neuropathic pain and atypical facial pain [11].
All patients recruited in the study were considered “end-of-stage algorithm patients” which means patients who have tried a pharmacological approach and invasive techniques without a lasting and concrete benefit. For invasive techniques, we refer to nerve blocks, radiofrequency of the GG or of the trigeminal branches, subcutaneous or peripheral nerve stimulation, and other techniques that are usually applied to face trigeminal neuropathic pain.
Surgical technique
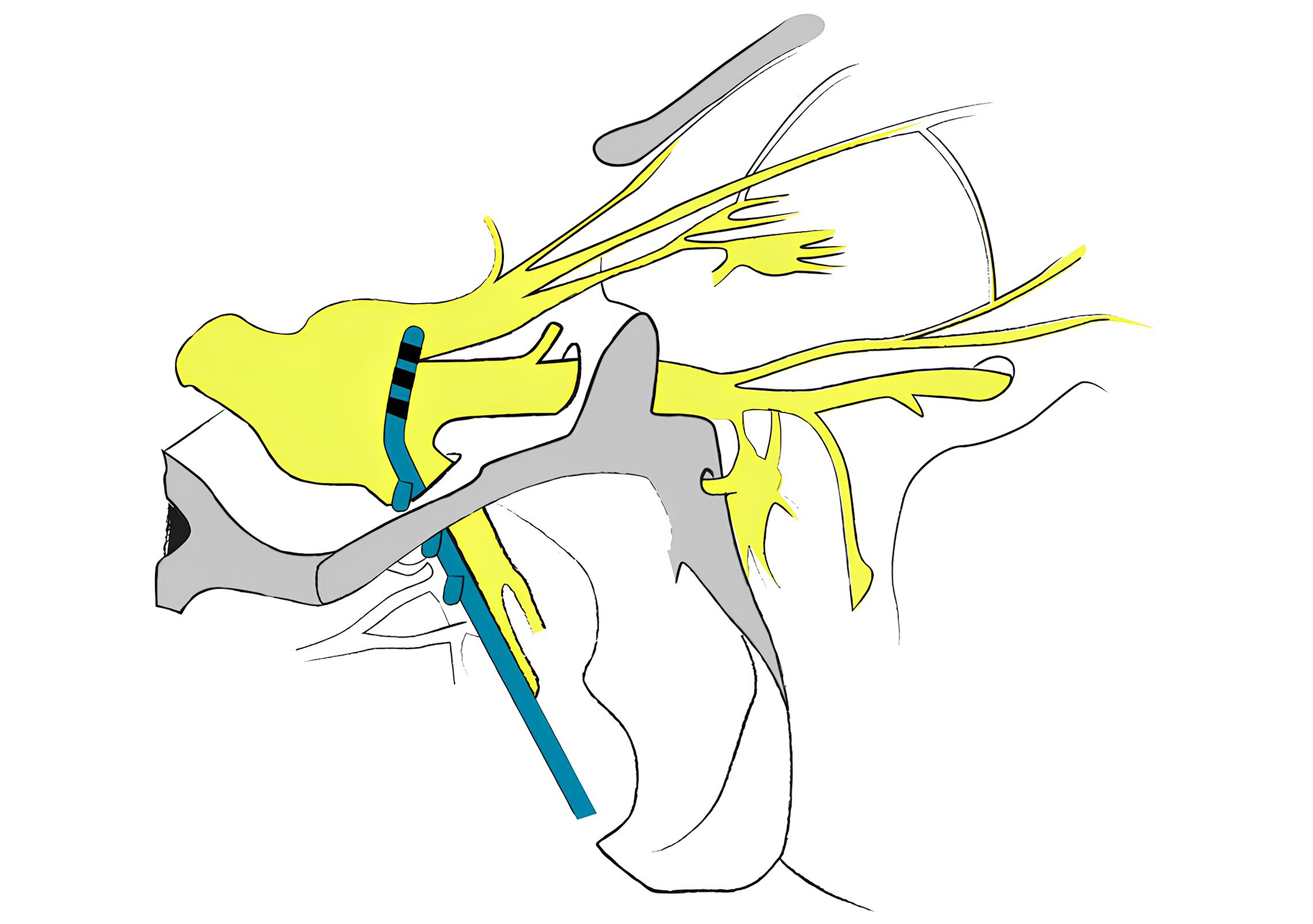
These patients were implanted with a custom-made tripolar, bent, tined lead, developed by Van Buyten in collaboration with Bakken Research Center Medtronic (Model 09053) (Figure 1). The lead is not commercially available worldwide and it has to be used off-label [compassionate use approved by the Federal Agency for Medicines and Health Products (FAMHP) of Belgium].

Medtronic lead model 09053. The three tines were developed to have a greater grip on tissues and prevent dislocation. Reprinted from “Logghe Y, Smet I, Jerjir A, Verelst P, Devos M, Van Buyten JP. Trigeminal neuropathy: Two case reports of gasserian ganglion stimulation. Brain Behav. 2021;11:e2379.”. CC BY. https://doi.org/10.1002/brb3.2379
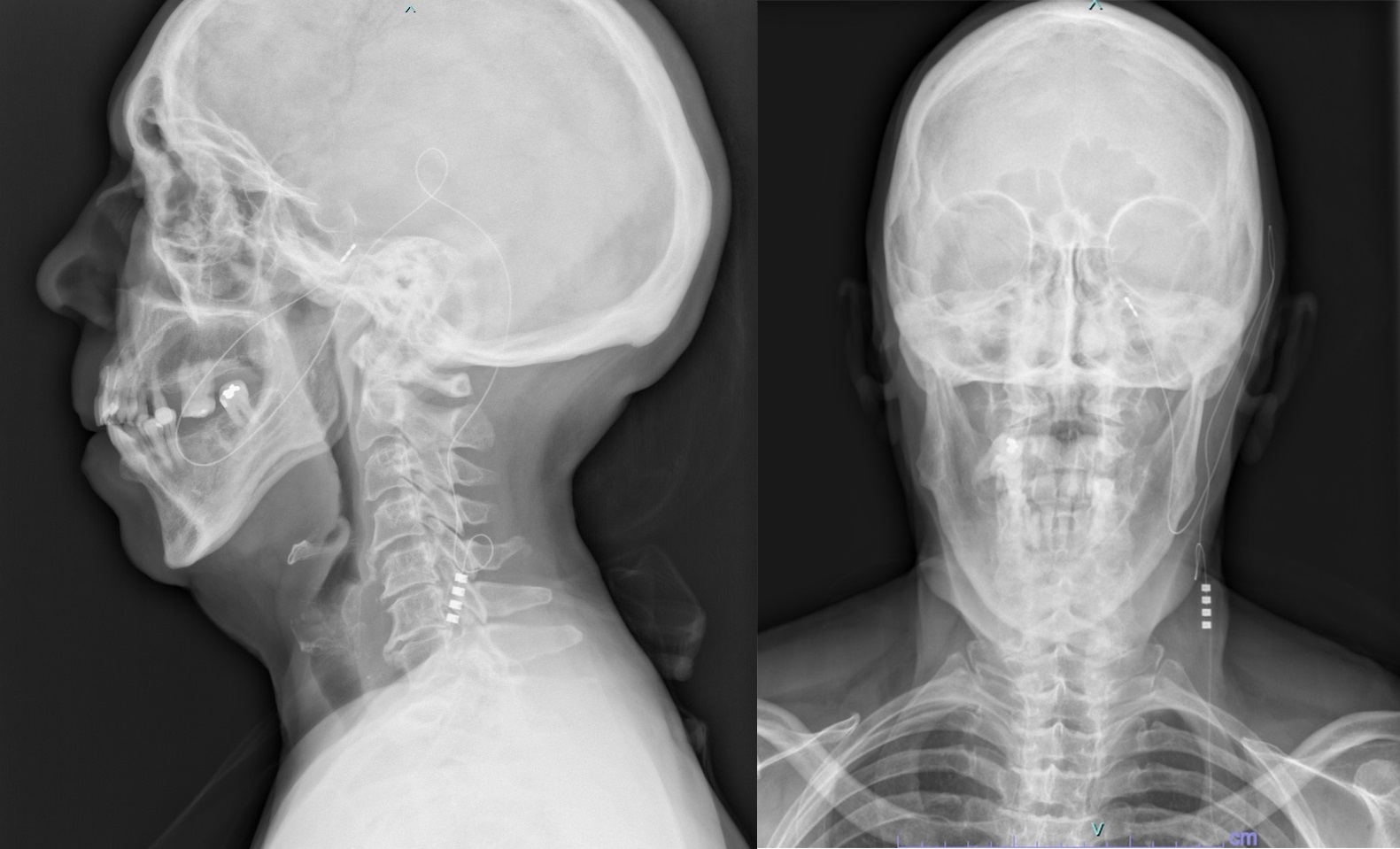
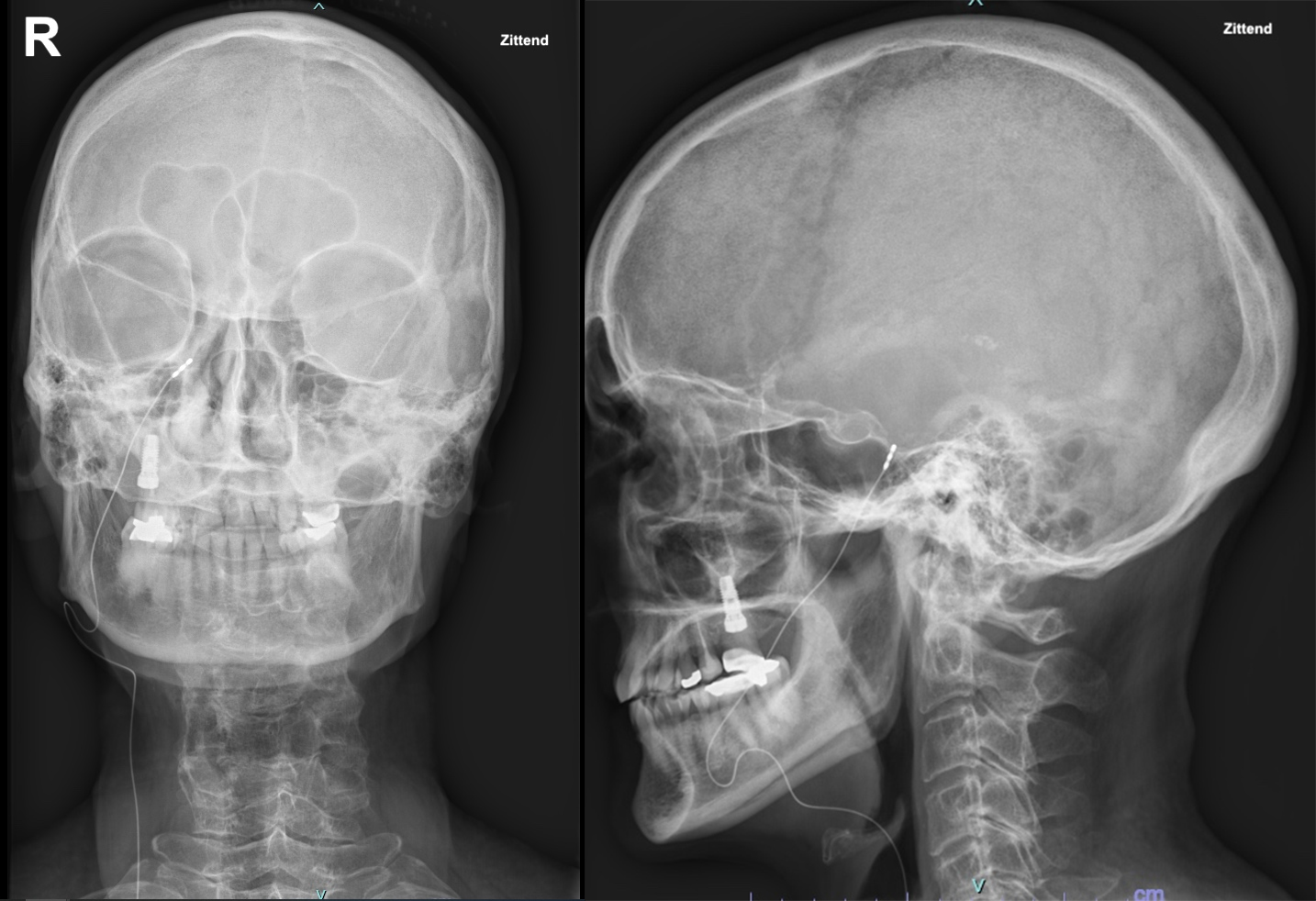
The procedure consists of a two-step surgery, the first of which is called the trial period. During the trial period, an electrode was placed into the foramen ovale near the GG using a percutaneous technique and connected to an external pulse generator. The procedure was performed under sedation, local anesthesia, and with antibiotic prophylaxis coverage. As a pre-treatment work-up, a head CT was acquired to guide needle placement into the foramen ovale using a coupled electromagnetic neuronavigational system. The needle, similar to those used for selective percutaneous thermocoagulation, was 15 gauges and was inserted about 2.5 cm lateral to the labial commissure. A small incision was made next to the entry point [12]. The needle was then guided by 3D real-time electromagnetic tip tracking system (Medtronic StealthStation™ S7 surgical navigation system) into the foramen ovale [13]. After passing through the foramen, the needle was advanced further until the tip had reached the clivus level. At this point, the electrode was inserted through the needle under continuous fluoroscopy until the tip also reached the level of the clivus (Figure 2).
The lead (Model 09053) had a diameter of 1.24 mm and until 2015 a length of 40 cm. Since 2016, the length has been changed to 70 cm. Then, a stimulation test was performed, and the electrode was gently moved to get a correct response from stimulation and a correct imaging position. Once a satisfactory position had been achieved, the needle was withdrawn under fluoroscopic control and the electrode was secured with stitches at the entry point. While the electrode was secured with a suture, the patient was deeply sedated for added comfort. From 2010 to 2015, the lead was tunneled behind the ear up to the neck. Thus, the electrode was then tunneled subcutaneously between the maxilla and mandibular region, and a second suture was made in this preauricular position. A third small incision in the upper portion of the neck was made where the lead was tunneled [12]. Here a temporary extension cable was connected to the lead and secured in this small subcutaneous pocket. The temporary extension cable has now been connected to an external pulse generator. From 2016 to 2022, the lead was inserted by passing through the insertion site and down directly through the neck using a tunneling tool. A pocket was created at the infraclavicular level where the connection between the electrode and the extension cable was placed. The extension cable was then connected to an external pulse generator (Figure 3 and Figure 4). The day after the trial implant patients were discharged, and an external battery was reprogrammed to be able to administer the most suitable therapy. In addition, during the trial period, the patients had a remote controller which allowed them to adjust the stimulation parameters. A confirmatory imaging of the correct lead positioning was obtained before discharge. The trial period was approximately one month for each patient if no complications occurred. Four patients were implanted without an external battery trial period because they had already an implanted battery for other neurostimulation therapies. If the trial period was successful, a permanent implant was performed, and the second step of the surgery was performed, this time under general anesthesia for better patient tolerance. An internal pulse generator (IPG) was inserted into a subcutaneous pocket made in the patient’s body and connected with an extension cable. The previous small subcutaneous pocket, where the provisional external extension cable was connected to the lead, was reopened, and the extension cable was removed. At this point, an internalized extension wire was connected to the lead and tunneled up to the pocket, which was made in the infraclavicular fossa. In two patients the battery was placed in the abdominal region while in one patient on the flank. The reason was that two of these patients already had an implanted battery, while the other did not want to have the battery implanted in the infraclavicular region.
Statistical analysis
The sample size was chosen based on the available clinical data at our institution. Continuous variables were described as mean, standard deviation, medians, and interquartile range, while categorical variables were reported with absolute frequencies. Comparison between patients’ cohorts was performed with the Chi-square test or Fisher’s test, as appropriate. The Kaplan-Meier method was used to estimate permanent implant survival. Excel was used for descriptive statistical analysis and Jamovi was used for inferential statistics analysis.
Results
Our cohort consisted of 49 patients (32 women and 17 men) aged from 32 to 76 (mean 56 ± 21) undergoing GG stimulation for the treatment of chronic facial pain. Most patients complained of trigeminal pain on the right side (53% right vs. 43% left). Only two patients presented trigeminal neuropathic pain on both sides: one was implanted on both sides while the other didn’t undergo the procedure because he did not tolerate paresthesia. The trigeminal branch most often involved in pain symptoms was V3, with a prevalence of 79% of patients (Table 1). Post-traumatic trigeminal neuropathic pain was present in 28 patients, thus 56% of the cohort, while 12 patients suffered from trigeminal pain after dental procedures. A total of 24 patients developed trigeminal pain after interventional procedures and four after traumatic injury. Only one showed trigeminal neuropathy in a sclerosis syndrome, while only two had trigeminal neuropathic pain after Varicella Zoster (Table 2). A greater number of patients with non-traumatic pain etiology (16 out of 21) received a permanent implanted GG stimulator, as compared to those with traumatic pain etiology (19 out of 28). However, there was no statistically significant association between pain etiology and permanent implant (Yates’s Chi-squared test, p = 0.950).
Baseline characteristics (n = 49)
| Variable | Values |
|---|---|
| Age (years) ± IQR | 56 ± 21 |
| Female | 32 (65%) |
| Male | 17 (35%) |
| End stage algorithm | 49 (100%) |
| Laterality | |
| Left | 21 (43%) |
| Right | 26 (53%) |
| Bilateral | 2 (4%) |
| Pain distribution | |
| V1V2V3 | 18 (37%) |
| V2V3 | 9 (18%) |
| V1V3 | 1 (2%) |
| V1 | 1 (2%) |
| V2 | 9 (18%) |
| V3 | 11 (22%) |
IQR: interquartile range
Pain etiology (n = 49)
| Pain etiology | n (%) | Definitive implant n (%) |
|---|---|---|
| Post-traumatic trigeminal pain after surgery | 11 (22%) | 10 (90.9%) |
| Post-traumatic trigeminal pain after dental surgery | 12 (24%) | 7 (58.3%) |
| Idiopathic trigeminal neuropathic pain | 5 (10%) | 3 (60.0%) |
| Classical trigeminal neuralgia with concomitant continuous pain | 11 (22%) | 8 (72.7%) |
| Post-traumatic trigeminal pain after alcoholization of Gasserian ganglion | 1 (2%) | 0 (0.0%) |
| Trigeminal postherpetic neuralgia | 2 (4%) | 2 (100.0%) |
| Trigeminal neuropathic pain post stroke | 1 (2%) | 1 (100.0%) |
| Trigeminal neuropathic pain post radiotherapy | 1 (2%) | 1 (100.0%) |
| Trigeminal neuropathic pain in multiple sclerosis syndrome | 1 (2%) | 1 (100.0%) |
| Post-traumatic trigeminal pain (head and face trauma) | 4 (8%) | 2 (50.0%) |
In this study, only major complications were considered, with no inclusion of minor complications, such as those resolved with reprogramming.
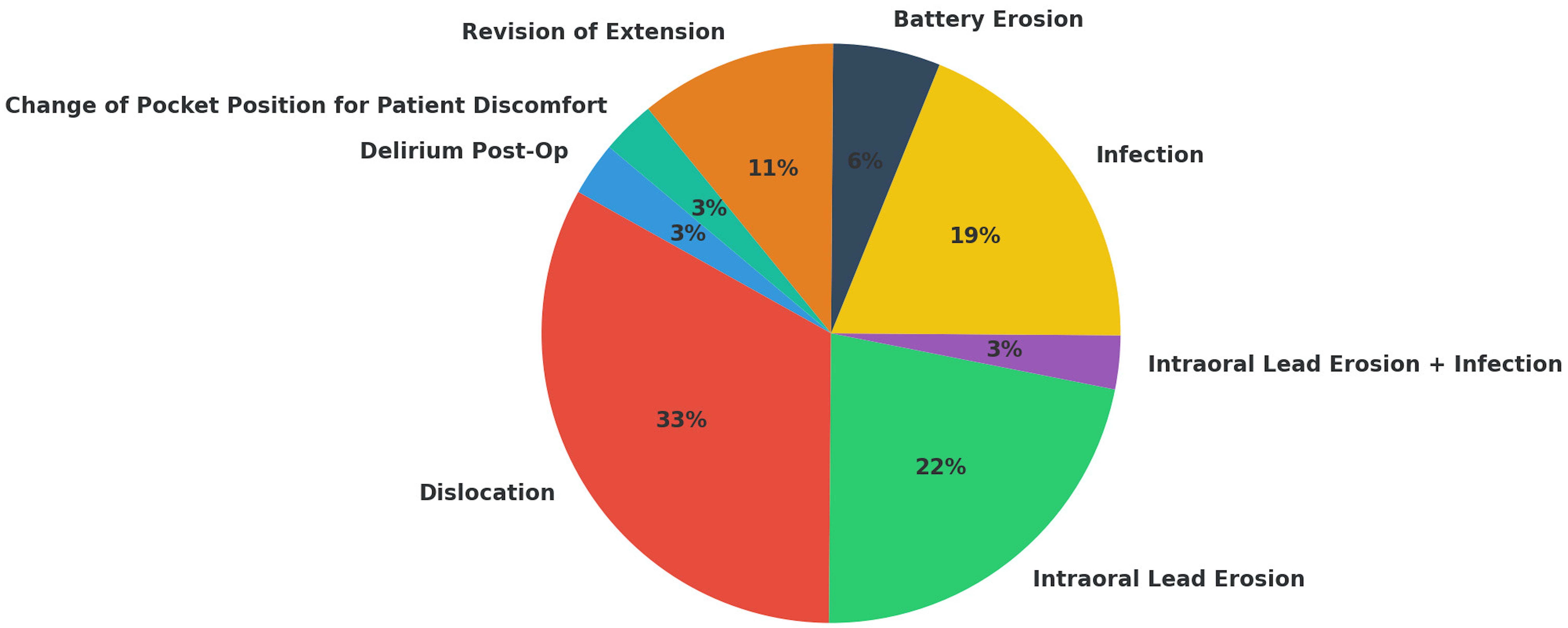
Over an average follow-up duration of three years, 36 complications were observed, with 23 patients experiencing at least one complication. Among these patients, only 39% experienced a second complication, and only one patient developed more than two complications. None of the complications resulted in permanent disability or death, and all of them were resolved. Conservative treatment resolved five complications, while surgery was necessary for the rest (Figure 5).
Comparison between the two different tunneling techniques
In our cohort of patients, we can identify two subgroups based on the tunneling technique used. A tunneling technique, which included passing the lead behind the ear, was used for 20 procedures. The insertion of 30 leads was instead carried out using a tunneling technique in which the lead was tunneled directly from the mandibula to the neck. We compared these techniques in terms of complications, and we saw that there is no statistically significant difference between the type of tunneling technique used and complications (Fisher exact test value 0.567, p = 0.451). Moreover, there is no statistically significant difference between the type of complications and the tunneling technique used (Fisher exact test, p = 0.181). The only noteworthy difference was in the group of the ear tunneling technique where we found a complication which is not present in the other group, the revision of the extension cable. This type of complication appears 4 times in the ear tunneling technique. Most complications occurred in the first year of follow-up in both groups (75th percentile 231–241 days) (Figure 6).

Box plot shows the correlation between time and the development of the first complication in our cohort of patients
Permanent implantation
Out of 50 trials of GG stimulation, 35 ended with the permanent implant. The rate of successful trial stimulation reported in our study (70%) follows previously published data, which ranges between 47%–80% [14]. Considering only those patients who underwent IPG implantation, we can observe that 11% of patients had electrode explants within one year of IPG implant, another 11% of patients in the second year, another 6% of patients in the third year, and only one patient (corresponding to 3%) in the fourth year (Figure 7).
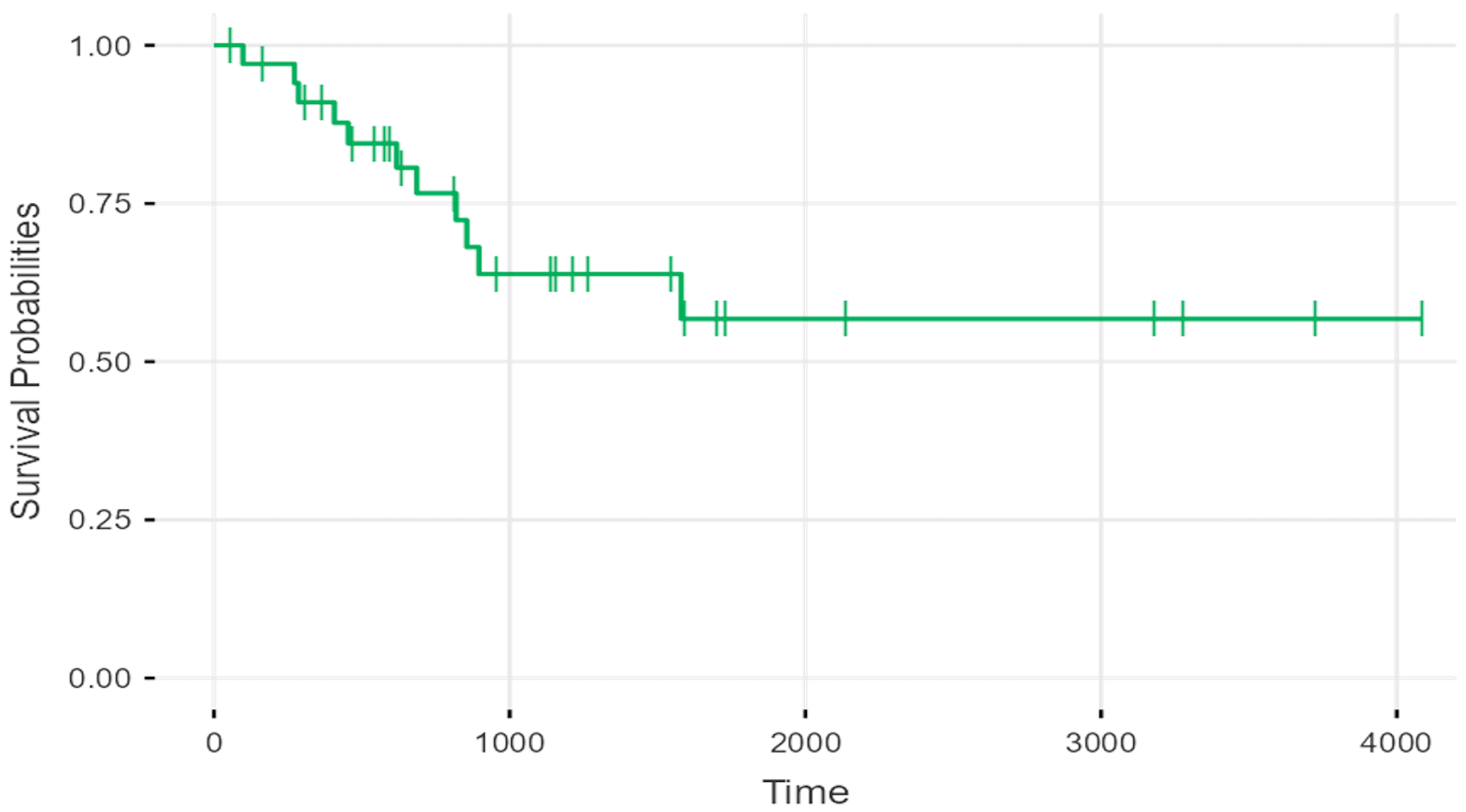
Kaplan Meier survival curve shows the trend of 35 patients with definitive implantation over time. The event considered is the implant removal. On the x-axis, time is expressed in days
Discussion
There are three well-established percutaneous procedures for the treatment of trigeminal pain: percutaneous balloon compression (PBC), radiofrequency thermocoagulation (RF), and percutaneous retrogasserian glycerol rhizotomy (PRGR). These techniques have been widely used for trigeminal neuralgia, yet their application extends to other forms of trigeminal pain as well, and all three methods demonstrate good outcomes and are considered safe [15]. However, when these techniques do not achieve a satisfactory benefit, GG stimulation may be considered a viable option. Although there is no randomized trial of GG stimulation in the literature, there are several papers showing its effectiveness in treating trigeminal pain [16–18]. In addition, there is no evidence in the literature on which tunneling technique is to be considered the gold standard and whether there is an association with the occurrence of complications technique-related. Our retrospective study shows that there are no statistically significant correlations between the tunneling technique used and the occurrence of complications nor does there appear to be any correlation between the type of tunneling technique used and the type of complication that occurred. However, it was found that in the group of patients in which the tunneling technique of passing the electrode behind the ear was used, a complication arose that did not occur in the other group of patients. This complication was the development of both skin erosion and some sort of skin fibrosis of the neck which produced discomfort for the patient. It is probably because the area behind the ear is very sensitive due to the small thickness of the skin. Also, connecting the electrode extension cable and the electrode itself in this delicate region can lead to fibrosis of the skin and subcutaneous tissues causing pain. This area is also very often subjected to additional pressure in patients using glasses and audio devices [2]. In all four patients with this complication, surgery was required to perform a revision of the extension cable.
If we consider the entire cohort of patients, we see that the most frequent complication is electrode dislocation. In our center, we only implanted the custom-made tripolar, bent, and tinned leads for the stimulation of the GG in order to reduce the frequency of dislocation. Despite this, dislocation resulted in 33% of total complications, and it was necessary to reposition the electrodes in all patients who developed this complication. A specific fixation system at the entry point could potentially decrease the dislocation rate. Mehrkens and Steude [19] demonstrated that lead size affects the incidence of displacement and the rate of bothersome dysesthesia. Their general impression was that larger leads appear to be less prone to dislocations but have a greater chance of causing dysesthesia. They reported discomfort and paresthesia induced by the size of the electrode from a diameter of 0.9 mm. Conversely, they noted that an electrode with a smaller diameter, i.e., 0.7 mm, was more prone to dislocation [19]. In our cohort of patients, we observed no unpleasant dysesthesia with stimulation despite the use of a 1.2 mm diameter lead. Another common complication in our patient cohort was intraoral erosion of the electrode. This complication depends mainly on the anatomical properties of the patient’s cheek, as a minimum mucosal thickness is necessary. We believe that an ultrasound evaluation of the patient’s cheek thickness may be crucial in determining whether or not to proceed with electrode implantation. In our cohort, it has been observed that electrode removal was required in 5 patients due to complications, which, in four of these, were intraoral erosion of the electrode. Infection accounts for 19% of total complications, and, in one patient, intraoral electrode erosion and infection were combined. We do not know whether thorough oral hygiene and oral antibiotic prophylaxis strategies can further reduce the occurrence of this complication. No severe infections such as meningitis or sepsis were encountered. Despite the tunneling technique used, we observed that about 70% of the complications occurred in the first year, so we believe that closer follow-up during the first year after implantation can allow for early identification and resolution of possible complications. Texakalidis et al. [20] found that patients with a history of trauma (facial/head trauma or surgery) had statistically lower odds for a successful trial, compared to others for pain etiology. In our study, we found the same tendency, even though we didn’t find any statistically significant association [20]. There is no comforting data in the literature regarding the efficacy of neuromodulation on postherpetic trigeminal neuropathic pain, and for this reason, it has been questioned whether it was a correct indication for neurostimulation [21–23]. Although there are only two cases of postherpetic trigeminal neuropathic pain in our patient cohort, both have been permanently implanted with good results, and one of them has been using GG electrostimulation for more than ten years.
Limitations
The results of the present study should be interpreted in the context of several limitations. First, it is a single-center study. Second, it is an observational retrospective, non-randomized study. Also, as in all retrospective studies, some patients were lost to follow-up. The results of the present study cannot be generalized, and our findings should be considered valid only for the electrode described.
Conclusions
GG stimulation is a safe technique. No patient suffered permanent disability or death. In the event of therapeutic failure, the electrode can be removed, and the anatomy of the patient’s body reverts to that prior to implantation. Despite this, the development of new technologies and greater patient selection are needed to reduce the complication rate, which is still high. Electrode dislocation remains the most frequent complication, and the development of a new electrode anchoring system would be desirable. Both tunneling techniques were found to be safe, but the technique that involves direct passage into the neck does not lead to fibrosis and erosion of the neck tissues as a complication; therefore, we believe the latter is more suitable.
Future studies with larger case series and sample randomization are also mandatory to consolidate our considerations and extend the results for other electrodes as well.
Abbreviations
| CT: | computed tomography |
| GG: | Gasserian ganglion |
| IPG: | internal pulse generator |
Declarations
Author contributions
EP: Conceptualization, Investigation, Data curation, Writing—original draft, Writing—review & editing. AJ: Conceptualization, Writing—review & editing. IS: Supervision. MD: Resources. PP: Formal analysis. JPVB: Supervision. All authors read and approved the submitted version.
Conflicts of interest
Materials for the procedure are provided by Medtronic Bakken Research Center, who developed a custom-made lead in collaboration with our department (Multidisciplinary pain center, VITAZ, Sint-Niklaas, Belgium). The authors declare that they have no other conflicts of interest.
Ethical approval
The local Ethical Committee of Vitaz (Sint-Niklaas, Belgium) approved the study (EC22022).
Consent to participate
Not required.
Consent to publication
Not required.
Availability of data and materials
The raw data supporting the conclusions of this manuscript will be made available by the authors (Edoardo Piacentino, piacentinoedoardo@gmail.com), without undue reservation, to any qualified researcher.
Funding
Not applicable.
Copyright
© The Author(s) 2025.
Publisher’s note
Open Exploration maintains a neutral stance on jurisdictional claims in published institutional affiliations and maps. All opinions expressed in this article are the personal views of the author(s) and do not represent the stance of the editorial team or the publisher.