-
 Special Issue Topic
Special Issue TopicBreast Cancer: Basic and Clinical Advances
Submission Deadline: December 31, 2023Guest Editor
Dr. Andrea Nicolini E-Mail
Department of Oncology, Transplantations and New Technologies in Medicine, University of Pisa, Pisa, Italy
About the Special Issue
Breast cancer is the most common malignancy worldwide and in females, with a social impact that underlines the relevance of any advances in basic and clinical research. In the last decades, much progress has been made in the comprehension of the molecular mechanisms sustaining tumor growth and diffusion, and certainly a large part of this research has been devoted to breast cancer. As a result of such intense investigation, many mechanisms have been elucidated, and a few targeted therapies through translational research have been introduced into current clinical practice. This is also named as "personalized" or "precision" medicine. This special issue aims to collect any useful scientific contributions in the breast cancer research field , either research or review articles that provide readers with information easy to catch and at the same time in line with the currently evolving studies.
Keywords: breast cancer, basic research, clinical research, molecular mechanisms, chemotherapy, immunotherapy
Published Articles
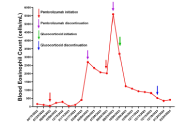 Pembrolizumab associated hypereosinophilia in locally advanced triple-negative breast cancerOpen AccessCase ReportThe advent of immunotherapy has revolutionized the therapeutic landscape of breast cancer. The immune checkpoint inhibitor drug, pembrolizumab, a monoclonal antibody targeting programmed cell death [...] Read more.Remo Poto ... Gilda VarricchiPublished: March 13, 2025 Explor Med. 2025;6:1001294
Pembrolizumab associated hypereosinophilia in locally advanced triple-negative breast cancerOpen AccessCase ReportThe advent of immunotherapy has revolutionized the therapeutic landscape of breast cancer. The immune checkpoint inhibitor drug, pembrolizumab, a monoclonal antibody targeting programmed cell death [...] Read more.Remo Poto ... Gilda VarricchiPublished: March 13, 2025 Explor Med. 2025;6:1001294
DOI: https://doi.org/10.37349/emed.2025.1001294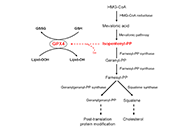 Association between GPX4 and HMGCR gene expression and cell proliferation in atypical hyperplasia of the breastOpen AccessOriginal ArticleAim: Using a dataset available from the NCBI Gene Expression Omnibus Repository, this in silico study investigated the differential expression of GPX4, the gene coding for the detoxifying enzyme [...] Read more.Danila CoradiniPublished: December 15, 2024 Explor Med. 2024;5:950–959
Association between GPX4 and HMGCR gene expression and cell proliferation in atypical hyperplasia of the breastOpen AccessOriginal ArticleAim: Using a dataset available from the NCBI Gene Expression Omnibus Repository, this in silico study investigated the differential expression of GPX4, the gene coding for the detoxifying enzyme [...] Read more.Danila CoradiniPublished: December 15, 2024 Explor Med. 2024;5:950–959
DOI: https://doi.org/10.37349/emed.2024.00268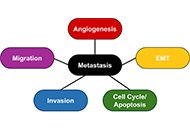 Role of transcription factors in metastasis of breast cancerOpen AccessReviewMetastasis causes a majority of deaths in breast cancer patients. Metastasis is the spread of cancer to distant sites in the body away from the primary tumor, creating secondary tumors, or metastase [...] Read more.Spoorthi Marada ... Yi LuPublished: December 09, 2024 Explor Med. 2024;5:936–949
Role of transcription factors in metastasis of breast cancerOpen AccessReviewMetastasis causes a majority of deaths in breast cancer patients. Metastasis is the spread of cancer to distant sites in the body away from the primary tumor, creating secondary tumors, or metastase [...] Read more.Spoorthi Marada ... Yi LuPublished: December 09, 2024 Explor Med. 2024;5:936–949
DOI: https://doi.org/10.37349/emed.2024.00267 Navigating breast health: a comprehensive approach to atypical ductal hyperplasia of the breast management and surveillanceOpen AccessMini ReviewAtypical ductal hyperplasia (ADH) is a benign lesion of the breast that is associated with an increased risk of invasive breast cancer. This review explores the pathophysiology, risk factors for pro [...] Read more.Nadia Islam, Suneela VeguntaPublished: February 07, 2024 Explor Med. 2024;5:59–64
Navigating breast health: a comprehensive approach to atypical ductal hyperplasia of the breast management and surveillanceOpen AccessMini ReviewAtypical ductal hyperplasia (ADH) is a benign lesion of the breast that is associated with an increased risk of invasive breast cancer. This review explores the pathophysiology, risk factors for pro [...] Read more.Nadia Islam, Suneela VeguntaPublished: February 07, 2024 Explor Med. 2024;5:59–64
DOI: https://doi.org/10.37349/emed.2024.00205 Cholesterol de novo biosynthesis: a promising target to overcome the resistance to aromatase inhibitors in postmenopausal patients with estrogen receptor-positive breast cancerOpen AccessOriginal ArticleAim: Cholesterol is an essential component of cell membranes and serves as a precursor for several bioactive molecules, including steroid hormones and isoprenoids. Generally supplied by the blood [...] Read more.Danila Coradini, Federico AmbrogiPublished: December 29, 2023 Explor Med. 2023;4:1079–1093
Cholesterol de novo biosynthesis: a promising target to overcome the resistance to aromatase inhibitors in postmenopausal patients with estrogen receptor-positive breast cancerOpen AccessOriginal ArticleAim: Cholesterol is an essential component of cell membranes and serves as a precursor for several bioactive molecules, including steroid hormones and isoprenoids. Generally supplied by the blood [...] Read more.Danila Coradini, Federico AmbrogiPublished: December 29, 2023 Explor Med. 2023;4:1079–1093
DOI: https://doi.org/10.37349/emed.2023.00196 The differential effect of the immune system in breast cancerOpen AccessReviewCancer cure with immunotherapy is an innovative step towards cancer treatment with better survivability, but it is mostly dependent on the response of the patient’s immune system to the immunother [...] Read more.Banashree Bondhopadhyay ... Vishakha KasherwalPublished: December 29, 2023 Explor Med. 2023;4:1094–1108
The differential effect of the immune system in breast cancerOpen AccessReviewCancer cure with immunotherapy is an innovative step towards cancer treatment with better survivability, but it is mostly dependent on the response of the patient’s immune system to the immunother [...] Read more.Banashree Bondhopadhyay ... Vishakha KasherwalPublished: December 29, 2023 Explor Med. 2023;4:1094–1108
DOI: https://doi.org/10.37349/emed.2023.00197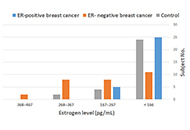 Impact of the type of breast cancer on the biodiversity of the vaginal Candida represented by estrogen receptor and its levelsOpen AccessOriginal ArticleAim: Estrogen has an important role in the colonization of Candida through the presence of estrogen receptors (ERs). These ERs are usually used to categorize breast cancer into two types, positiv [...] Read more.Ali Abdul Hussein S. AL-Janabi ... Abdul Razzak Kalaf HassanPublished: December 06, 2023 Explor Med. 2023;4:878–885
Impact of the type of breast cancer on the biodiversity of the vaginal Candida represented by estrogen receptor and its levelsOpen AccessOriginal ArticleAim: Estrogen has an important role in the colonization of Candida through the presence of estrogen receptors (ERs). These ERs are usually used to categorize breast cancer into two types, positiv [...] Read more.Ali Abdul Hussein S. AL-Janabi ... Abdul Razzak Kalaf HassanPublished: December 06, 2023 Explor Med. 2023;4:878–885
DOI: https://doi.org/10.37349/emed.2023.00183 -
-
Ongoing Special Issues
-
Completed Special Issues