Abstract
Aim:
A Zika virus outbreak that began in Brazil, developed into an international public health emergency that extended from February 2015 until November 2016. Zika-infected pregnant women gave birth to a cohort of infants with congenital Zika syndrome (CZS) originally defined by severe microcephaly, retinal scarring, joint deformities, and hypertonia. This study examines the nature, extent, and severity of all CZS clinicopathologic findings described to date, compiled and analyzed by system. It reviews studies monitoring disease progression and proposing classification schemes for CZS stages. The teratogenic cellular and molecular mechanisms implicated in CZS pathogenesis are also discussed.
Methods:
A systematic review was conducted by literature search through WorldCat.org and ProQuest Central databases to identify studies on case series from the 2015–2016 CZS outbreak.
Results:
Twenty-six reports were included describing radiologic, ophthalmologic, audiologic, orthopedic, and laboratory test results in CZS cases including stillborns between 2016 and 2023. CZS neuropathology included prenatal and postnatal microcephaly, cerebral calcifications, quadriparesis, epilepsy, ventriculomegaly, reduced cerebral parenchyma, malformation of cortical development, and sleep electroencephalogram disturbances. Visual deficits were due to retinal and optic nerve lesions. Conductive and sensorineural hearing deficits were stable. Hypertonia, hypotonia, and spasticity with foot, hip, knee, and shoulder deformities resulted in arthrogryposis and restricted joint mobility. There was enlargement of immune organs, increased leukocyte counts, and cytokine dysregulation. Oro-craniofacial deformities affected the midface and caused dental eruption delay. Additional studies proposed that these systemic teratogenic effects could be attributable to transplacental Zika virus infection of multiple fetal progenitor cell lineages.
Conclusions:
The CZS-associated impairments in brain, eye, musculoskeletal, and immunologic functions caused disabilities that varied from moderate to severe, and significantly increased age-specific mortality rates. Further research is warranted to assess progression, classify stages, elucidate the precise molecular mechanisms mediating Zika teratogenicity, develop suitable therapeutic strategies, and design supportive social policies.
Keywords
Congenital Zika syndrome, teratogenic, microcephaly, cerebral palsy, epilepsy, craniofacial, arthrogryposis, immune dysregulationIntroduction
Zika virus infection is a mosquito-borne disease transmitted by infected Aedes aegypti and Aedes albopictus mosquitoes. Zika is a single-stranded RNA virus of the Flaviviridae family that was first discovered in Uganda in 1947 in rhesus monkeys (Macaca mulatta), and the first human cases were detected in 1952 by serology [1]. In 2007, the first outbreak occurred in the Pacific islands. The 2015–2016 Zika outbreak was first reported in Brazil as it spread from countries in South America including Columbia, through Central America, the Eastern Caribbean, US Virgin Islands, with widespread transmission through Puerto Rico, and local spread into Florida and Texas within the United States. In February of 2015, the World Health Organization (WHO) declared Zika virus a public health emergency of international concern, a status that continued until November of 2016 [1]. However, women who were pregnant while exposed to Zika virus in 2016 delivered their babies in 2017.
Zika febrile illness symptoms appeared 3–14 days after initial infection and included mild fever, rash, conjunctivitis, headache, muscle pain, joint pain, abdominal pain, and fatigue. In addition to mosquito bites, Zika virus could be transmitted through blood transfusions and other body fluids. Increased rates of miscarriages and microcephalic newborns prompted the Center for Disease Control and Prevention (CDC) to declare in April 2016 that Zika virus was a teratogenic microbe associated with a pattern of anomalies resulting in neurodevelopmental, ophthalmologic, and neuromuscular dysfunction attributed to impaired fetal brain development [2]. According to the CDC, 1 in 10 Zika-infected pregnant women delivered babies with a phenotype of birth defects pathognomonic of the congenital Zika syndrome (CZS) [3].
Clinical observation revealed that Zika infection in-utero caused microcephaly with one or more significant complications including visual impairment, hearing loss, and articular or musculoskeletal abnormalities [4]. Intensive mosquito control was implemented to limit transmission in the USA due to the alarming concern of infants being born with profound neurologic deficits as a result of maternal infection with the Zika virus [5]. To further identify the precise neuropathologic and retinal lesions, orthopedic and otolaryngologic disorders, and underlying cellular mechanisms of this new congenital syndrome, studies have been evaluating neuroradiologic, histopathologic, audiology, neurodevelopmental as well as immunologic parameters. However, small sample sizes, diminishing population due to higher mortality rates, communication difficulties due to intellectual disability, confidentiality concerns, and loss to follow-up, as well as the coronavirus disease 2019 (COVID-19) pandemic have presented challenges to prioritizing and conducting such investigations on a global scale. As a result, there was a disappointing lack of comprehensive, large-scale worldwide, prospective cohort studies that could have been providing regular systematic updates on the outcomes and progress of those who have been living with this new syndrome for the last 8 years. Nevertheless, this paper compiles the available quantitative results by system, and analyzes the statistically significant clinicopathologic findings across studies. Overall, the quality of life was diminished to variable extents by the impact of more than 76 abnormalities, listed below, which effected neurodevelopmental delays, intellectual disabilities, susceptibility to infections, other health problems, and life-threatening complications [3].
In the Results section, the clinicopathologic findings are grouped according to the primary system investigated in the studies. The immunologic and central nervous system (CNS) abnormalities joint and musculoskeletal abnormalities, visual disturbances, retinal and optic nerve lesions, audiology, and oro-craniofacial defects are summarized. The definitions, CZS-associated pathophysiology and clinical sequelae for each of the identified abnormalities are summarized in the following list, ordered by organ system:
Brain pathology
Microcephaly: small head size with head circumference falling more than two standard deviations below normal; may be due to poor brain growth and decreased brain volume
Brain parenchymal atrophy: shrunken volume of brain white matter and gray matter due to loss of neurons
Ventricle enlargement: enlargement of the fluid-filled cavities within the brain (hydrocephalus, ventriculomegaly) due to maldevelopment of brain tissues around the ventricles; may be due to either loss of neurons causing atrophy, blockage in the flow of cerebrospinal fluid (CSF) from the brain to the spinal cord, or lack of absorption of CSF
Enlarged choroid plexus: villous hypertrophy of the choroid plexus (hyperplasia) in the ventricular lining is associated with overproduction of CSF
Agyria: lack of brain convolutions (gyri)
Pachygyria: decreased convolutions that are unusually thick producing a relatively smooth cerebral surface due to neuronal migration abnormalities in the developing brain
Lissencephaly: completely smooth brain surface with no convolutions due to severe neuronal migration abnormalities in the developing brain
Malformation of cortical development: disordered layers of neurons in the gray matter due to disturbed sequence of neuronal migration during cerebral cortex formation
Encephalomalacia: cavity in a necrotic (liquifying) area of the brain; may be due to infection or ischemia (hypoxia)
Dysgenesis of corpus callosum: small or absent corpus callosum portion of the brain that connects the two cerebral hemispheres
Subcortical calcifications: calcifications at the grey matter-white matter interface; the most common cause of intracranial calcifications are congenital infections
Enlarged subarachnoid space: may be associated with developmental delays
Destruction of cerebellar vermis: cerebellar dysfunction impairs balance and causes staggering gait, truncal ataxia, tremor of the head, uncoordinated movements, difficulties with speech (dysarthria), visual problems (nystagmus), and vertigo
Irritability and seizures: agitation and convulsions due to abnormal and uncontrolled electrical brain activity; may be triggered by any structurally abnormal focus in the brain
Abnormal sleep: electroencephalogram (EEG) shows absence of one or more of the five stages of sleep [wake, N1, N2, N3, and rapid eye movement (REM)]; may reflect abnormal neurophysiology
Spinal cord pathology
Wallerian degeneration in the spinal cord and brain stem: anterograde degeneration proceeding from the distal end of an axon following axonal injury
Immunology
Zika virus identified within fetal brain indicates fetal tissue infection with the virus
Activated microglia occurs in response to brain cell infection, injury, or damage
Enlarged lymphoid organs: benign hyperplasia of the thymus, spleen, and lymph nodes may be due to impaired immune functions resulting in increased demand, size, and weight
Fibrosis of lymphoid organ: scarring implies nonfunctional organ
Leukocytosis: increased counts of white blood cells may be due to increased demand for hematopoiesis to compensate for abnormal immune cell functions
Hypersegmented neutrophils: abnormal polymorphonuclear white blood cells with 6 or more lobes in their nucleus may be seen in congenital syndromes, vitamin B12, or folic acid deficiency
Increased count of atypical lymphocytes: may be due to rapid production and early release of relatively immature lymphocytes
Reduced cellular immune memory: weak cell-mediated immune responses
Cytokine level alterations: dysregulation of immune responses reflected by changes in levels of pro-inflammatory cytokines [increased interferon-gamma (IFN-γ) and interleukin-2 (IL-2), decreased IL-7)] and anti-inflammatory cytokines (increased IL-4)
Chemokine C-X-C motif chemokine ligand 10 (CXCL10), also known as IFN-γ-induced protein 10 kDa (IP-10), decreased levels which may decrease chemotaxis
Granulocyte-colony stimulating factor (G-CSF) level alteration: decreased G-CSF; may impair hematopoiesis
T lymphocytes throughout the brain may be due to lack of intact blood-brain barrier
Musculoskeletal and oro-craniofacial defects
Cerebral palsy: motor disability due to brain pathology limiting the use of muscles for moving and maintaining balance and posture
Motor milestones not achieved: motor function deficit due to neurodevelopmental delay
Hypertonia: abnormally high muscle tension due to upper motor neuron dysfunction; may be spasticity (stiffness), dystonia (spasms), or rigidity (inflexibility); may limit the ease of joint movement
Spastic tetraparesis: motor weakness in upper and lower limbs due to neurological degeneration
Low birth weight: term baby born weighing less than 2,500 g; associated with increased risk of birth defects and may be due to infection during pregnancy
Preterm birth: born prior to 37 weeks of gestation; associated with birth defects and may be due to maternal infection
Craniofacial disproportion: proportions of facial morphology (ears long and high-set, face broad and short, intercanthal distance and posterior nasal length short, nasal root prominent, nasal wings broad, mandibular retrognathia), and cranial vault shape (head circumference smaller, head height, head length, and naso-frontal angle) significantly different from normal controls
Frontotemporal retraction
Prominent occiput: increased convexity of the posterior skull
Biparietal depression: cranial depressions involving both parietal skull bones
Dolichocephaly: elongated head
Mesocephaly: intermediate length and width of the skull
Protruding maxilla: exposure of the upper teeth due to dental malocclusion
Hypoplasia of middle third of face: in midface hypoplasia, the eye sockets, cheekbones, and upper jaw do not grow as much as the rest of the face
Short lingual frenulum: the connection between the tongue and the floor of the mouth is shorter than usual; it may restrict tongue movement
Abnormal insertion of upper labial frenulum: problem with the connection between the upper lip and the upper gum; may be related to the presence of gaps between teeth (diastemas), may alter the growth of teeth, may expose the roots by pulling the gum away from teeth
Ogival palate: high-arched palate can lead to obstructive breath disorder, speech problems, sleep disruption, and sudden death
Retrognathic mandible: the lower jaw is positioned posterior to the upper jaw; may impair mastication and phonation; prognathic maxilla: upper jaw protrudes out of the face
Open bite: teeth are not aligned when the jaw is closed due to malocclusion; may interfere with chewing and biting ability, cause speech difficulties, and damage back teeth
Bruxism: grinding, clenching, or gnashing teeth while awake or asleep
Oligodontia: congenital absence of 6 or more teeth in primary and/or permanent dentitions due to anomalous dental development
Delay in dental eruption chronology or sequence is more common in congenital infections, premature births, and genetic factors
Enamel opacity: developmental defect of enamel; common in other congenital infections
Enamel hypoplasia: disturbed enamel production during the matrix formation stage; may cause sensitivity and increase risk of cavities
Joint deformities
Arthrogryposis: multiple joint contractures; joint stiffness limits its range of motion; may be due to underdeveloped (hypoplastic) limb muscles; limited shoulder and hip abduction, limited knee extension, limited foot flexion
Hip displacement (dysplasia): asymptomatic in early stages; progressive proximal femoral deformities and hip socket (acetabular) dysplasia (too shallow to cover the femoral head); hip abnormal acetabular index; due to a combination of hip muscle spasticity, weakness, and delayed weight-bearing
Hip dislocation: unstable hip joint in which the femoral head might be pushed out of the acetabulum (socket); high hip femoral head migration and subluxation; may be congenital due to developmental dysplasia of the hip
Foot deformities: clubfoot, syndactyly
Knee deformity
Shoulder internal rotation: excess nuchal skin
Swan neck deformity of fingers
Ophthalmologic complications
Chorioretinal scar: a geographic area of fibrosis in the choroid and retina; may be postinfectious; macular chorioretinal atrophy/scarring impairs vision
Retinal pigment epithelial mottling: abnormal development of pigment blotches in the pigment epithelium of the retina; may impair vision
Fovea reflex loss: absence of foveal reflex may be due to macular abnormalities; absence of foveal pit may be due to prematurity
Retinal vessel attenuation: vasoconstriction due to decreased metabolic demand following retinal photoreceptor degeneration
Optic nerve increased cup-to-disk ratio and pallor: non-glaucomatous optic nerve cupping with pallor of the residual neuroretinal rim; may lead to loss of visual acuity
Optic nerve hypoplasia: congenital underdevelopment of one or both optic discs with reduced number of optic nerve axons; may result in visual impairment or blindness
Iris coloboma: defect in the iris due to missing tissue
Lens subluxation: partially displaced lens
Exotropia: the eye is deviated outward; this may be due to 3rd cranial nerve palsy
Esotropia: the eye is deviated inwards towards the nose due to uncoordinated orbital muscles; can cause permanent vision loss if not corrected
Strabismus: hypertropia (crossed eyes, misaligned), may be due to uncoordinated orbital muscles, cranial nerves palsies, or brain lesions in centers that direct eye movements
Nystagmus: uncontrollable movements of the eye causing blurry and jumping vision; can be horizontal, vertical, or rotary
Astigmatism: distorted image focus due to irregular corneal shape
Hyperopia: farsightedness due to the image becoming focused behind the retina
Myopia: nearsightedness due to the image becoming focused in front of the retina
Audiologic complications
Conductive hearing loss: if due to incomplete formation of the external ear, it may be due to the maldevelopment of the first and second branchial arches and the first branchial cleft. If due to a defect in the middle ear ossicles, it can be due to otosclerosis, cholesteatoma, or otitis media with effusion
Sensorineural hearing loss: deafness from damage to the inner ear due to dysfunction of the cochlear hair cells and/or the auditory nerve (8th cranial nerve)
This study provides neonatologists, ophthalmologists, audiologists, pediatric neurologists, neurogeneticists, learning disabilities specialists, disabilities services officers, educational therapists, obstetricians, orthopedists, physical therapists, neuropsychologists, neuropathologists, microbiologists, immunologists, experimental biologists, and public health and health policy makers with an appreciation of (a) the multifaceted healthcare services needed to support families of children with CZS, (b) the urgency of needed preventive measures to protect against CZS in future outbreaks, and (c) the scientific initiatives needed to investigate the molecular mechanisms of Zika teratogenicity.
Materials and methods
A systematic review was conducted by literature search through ProQuest Central and WorldCat.org, inclusive of PubMed and EBSCOhost electronic databases. Additional resources were accessed through the CDC, WHO, and the Cochrane Library. The text words “Zika virus”, “Zika infection”, and “Zika epidemic” with the use of the Boolean operator “AND”, “congenital Zika syndrome”, “microcephaly”, “neuropathology”, “craniofacial”, and “immunology” were used to identify relevant studies discussing the clinical and pathological manifestations of CZS in the cohort of children who were born to mothers infected by the Zika virus during pregnancy.
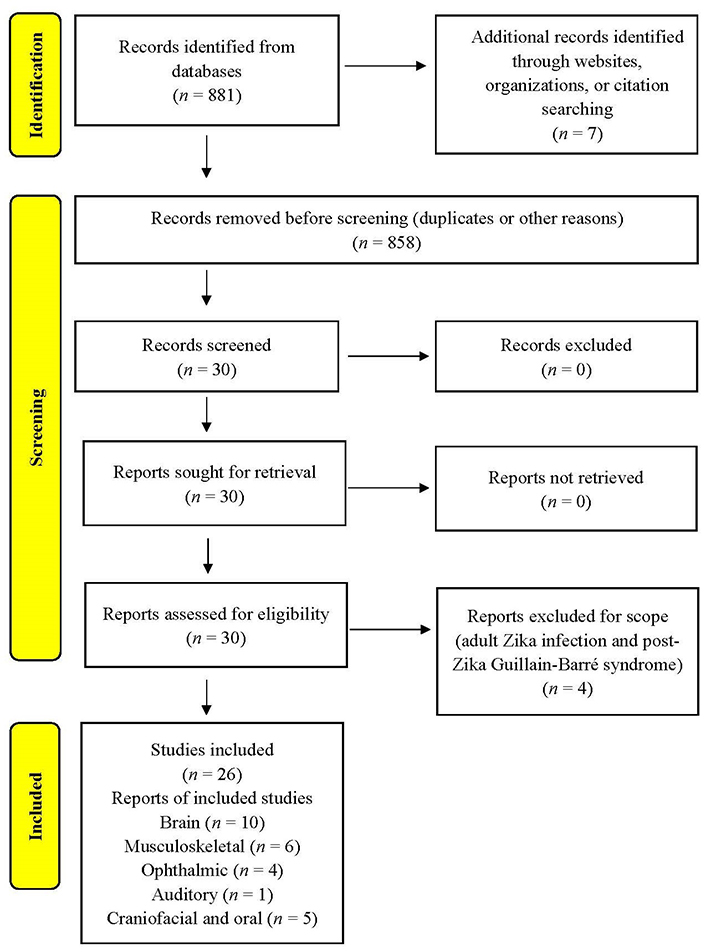
Inclusion criteria were 1) must be a scholarly or peer-reviewed source, 2) a relevant article published since 2015, and 3) articles available in the English language. Exclusion criteria were the following: 1) publications potentially used for marketing purposes, 2) articles earlier than 2015, and 3) articles not available in English. The search strategy is presented as a PRISMA flow diagram in Figure 1.
Data extraction was performed by 3 reviewers. Articles were rated according to the following scheme: (1) properly conducted randomized controlled clinical trial (RCCT), (2) well-designed controlled trial without randomization, or prospective comparative cohort studies, (3) case-control studies, or retrospective cohort study, (4) cross-sectional study, and (5) case report or series.
Results
Altogether 30 articles were identified and screened. After exclusion of 4 records, 26 studies on congenital Zika cases were included in synthesis. The reports were from 2016 (n = 5), 2017 (n = 3), 2018 (n = 1), 2020 (n = 2), 2021 (n = 6), 2022 (n = 7), and 2023 (n = 2).
The studies reported on fetal and neonatal findings as well as findings in older pediatric age groups. CNS and immunologic findings were systematically tabulated in Table 1, joint and musculoskeletal abnormalities in Table 2, vision impairment and eye pathology in Table 3, hearing impairments in Table 4, and craniofacial and oral alterations in Table 5.
CNS and immune system manifestations in CZS
| Study | Demographics | Findings |
|---|---|---|
Salmeron et al. 2022 [6] Cross-sectional case-control study | • 22 CZS cases diagnosed at birth in Brazil (n = 22); negative for Zika virus in the blood • 20 healthy controls • Age range 9 months to 5.4 years; average age 35.8 months ± 19.2 months • Females (n = 21), males (n = 21) | • Microcephaly in CZS cases (n = 22) • Morphological alterations in lymphoid organs:
• Leukocytes (WBC): increased count (46%, n = 6) • Eosinophils: increased count (62%, n = 8) • Lymphocytes: increased count (23%, n = 3), increased atypical morphology (P < 0.005), decreased normal morphology (P < 0.005) • Segmented neutrophils: increased count (15%, n = 2), increased number hypersegmented (P < 0.0001), increase hypersegmentation + cytoplasmic vacuolation (P = 0.019), decreased normal morphology (P < 0.0001) • Monocytes: increased count (23%, n = 3) • Cytokines: increased IFN-γ (P = 0.017), IL-2 (P = 0.017), IL-4 (P = 0.018), IL-5 (P = 0.022) • Reduced cellular immune memory: tuberculin skin test nonreactive (57%, n = 4), weakly reactive (43%, n = 3) in Bacillus Calmette-Guérin (BCG) vaccinated |
Rua et al. 2022 [7] Prospective cohort study | • Cohort of 38 children • Median age 4.3 months; examined at 4, 8, 12, 18, and 24 months of age • Females (n = 11), males (n = 27) | • Microcephaly (79%, n = 30) • Cranial (magnetic resonance imaging) MRI scan: malformation of cortical development and brain parenchymal atrophy (82%), ventricle enlargement (76%), dysgenesis of corpus callosum (73%), subcortical calcifications (84%), T2-weighted fluid-attenuated inversion recovery (FLAIR): hyperintense periventricular signals suggestive of delayed myelination • Motor milestone not achieved (82%, n = 31) • Irritability 50% at 8 months, 27% at 24 months • Epilepsy (71%): spasms in the 1st year of life (52%), focal seizures in the 2nd year of life (50%) • Axial hypertonia 77% at 4 months, 50% at 24 months • Spastic tetraparesis (> 80%) |
Nunes et al. 2021 [8] Cross-sectional case series study | • Cohort of 43 microcephalic children from Brazil • Age range 13 months to 42 months • Females (n = 19), males (n = 24) | • Proposed mild, moderate, and severe classification scheme for neuroimaging abnormalities • Documented multiple types of seizures by EEG ictal and interictal activity • Early seizures in younger than 6 months (42%) • Abnormal EEG total (72%), during sleep (63%) • Only 56% (n = 24) were able to register wakefulness and sleep • No awake recording (21%, n = 9) registered only sleep • No sleep recording (23%, n = 10) registered only wakefulness |
Nascimento-Carvalho et al. 2021 [9] Cross-sectional case-control study | • 14 Zika virus-exposed neonates: 7 with microcephaly (50%) and 7 without microcephaly (50%) • 14 normal control neonates | • Perturbed inflammatory mediator profiles in CSF • Higher levels of IL-4 (P = 0.01) in cases with microcephaly (n = 7) as compared to normal controls • Lower levels of IL-1α (P = 0.004) in cases without microcephaly (n = 7) as compared to normal controls • Lower levels of IL-7 (P = 0.048), IP-10/CXCL10 (P = 0.03), and G-CSF (P = 0.047) in total Zika virus-exposed cases (n = 14) as compared to normal controls |
Cavalcante et al. 2021 [10] Prospective cohort study | • Cohort of 110 CZS cases • Age up to 36 months • Females (n = 45), males (n = 65) • Compared CZS outcomes in cases born without microcephaly (n = 32) vs. cases with microcephaly identified at birth (n = 61) | • Only slight differences in manifestations and outcomes between CZS cases with and without microcephaly; postnatal-onset microcephaly (87.5%, n = 28; severe in 65.6%, n = 23) • Case fatality rate: 5.5% (n = 6), main cause was severe CZS congenital anomaly and pneumonia • Low birth weight (22%), preterm birth (13.3%) • Craniofacial disproportion (90.6%), frontotemporal retraction (71.7%), prominent occiput (55.7%), biparietal depression (46.2%) • Brain calcifications (93.5%), ventriculomegaly (88.8%, P = 0.002), reduced cerebral parenchyma (85.8%, P < 0.001), malformation of cortical development (78.3%, P = 0.047) • Spasticity (97%) • Epileptic seizures (90.7%) • Chorioretinal scar (22.5%) • Focal retinal pigmentary mottling (24.5%) • Excess nuchal skin (24.5%) |
Štrafela et al. 2017 [11] Case report | • Stillborn 32-week gestation • Sex unknown | • Low brain weight (84 g) compared to age-matched control • Agyria and pachygyria in the cerebrum, cerebellum, brain stem • Microcalcifications in gray matter and cortical-white matter junction • Enlarged lateral ventricle in the parieto-occipital region • Large encephalomalacia cyst in the right occipital lobe • Scattered T lymphocytes throughout the brain |
Driggers et al. 2016 [12] Case report | • Terminated 21-week gestation • Sex unknown | • Decrease in fetal head circumference from 47th percentile at 16 weeks to 24th percentile at 20 weeks • Presence of flavivirus in serum at 16 weeks • Abnormal intracranial anatomy at 19 weeks • Diffuse atrophy of cerebral mantle at 20 weeks • Low brain weight 30 g (reference weight, 49 g ± 15 g) |
Mlakar et al. 2016 [13] Case report | • Stillborn 32-week gestation • Sex unknown | • Ultrasonography: intrauterine growth retardation, calcifications, microcephaly, ventriculomegaly, transcerebellar diameter below second percentile • 1,470 g bodyweight (5th percentile) • Almost complete agyria, open sylvian fissures, internal hydrocephalus of lateral ventricles • Activated microglial cells and some HLA-DR expressing macrophages throughout cerebral gray and white matter • Wallerian degenerations in brain stem and spinal cord • RT-PCR identified Zika virus in fetal brain sample |
Cavalheiro et al. 2016 [14] Cross-sectional case series study | • 13 microcephalic newborns • Sex unknown | • Craniofacial disproportion (100%, n = 13) • Craniofacial hypoplasia of corpus callosum (100%, n = 13) • Craniofacial lissencephaly (100%, n = 13) • Craniofacial increased subarachnoid space (100%, n = 13) • Intracranial calcifications (100%, n = 13) • Ventriculomegaly (100%, n = 13) • Enlarged choroid plexus (61.5%, n = 8) |
Sarno et al. 2016 [15] Case report | • Stillborn 32-week gestation • Female | • Zika RNA found in CNS tissues • Microcephaly, hydranencephaly, intracranial calcification, and posterior fossa with destruction of the cerebellar vermis at 26 weeks and 30 weeks gestation • Female fetus weighed around 930 g and had signs of microcephaly and arthrogryposis |
HLA-DR: human leukocyte antigen-DR; RT-PCR: reverse-transcriptase-polymerase-chain-reaction
Joint and musculoskeletal manifestations in CZS
| Study | Demographics | Findings |
|---|---|---|
Magalhães et al. 2023 [16] Retrospective cohort study | • Cohort of 29 CZS cases • 5-year follow-up • Age > 24 months • Females (n = 16), males (n = 13) | • Microcephaly (100%, n = 29) • Spastic quadriparesis (93.1%, n = 27) • Spastic hemiparetic (6.9%, n = 2) • Arthrogryposis (3.4%, n = 1) • Hip abnormal acetabular index under 2 years of age • High hip femoral head migration and subluxation over 2 years of age |
da Fonseca et al. 2022 [17] Prospective cohort study | • Cohort of 30 CZS cases • 2 pelvic radiographs performed within 12 months • Age range from 19 months to 43 months • Females (n = 11), males (n = 19) | • Microcephaly (87%, n = 26) • Optic nerve abnormalities and chorioretinal (53%, n = 30) • Spasticity, hip functional limitation, hip deformity in flexion, adduction, and medial rotation bilaterally (83%, n = 25) • Hip displacement (total 47%, n = 14; right hip 43%, n = 13, left hip 30%, n = 9) • Hip dislocation (20%, n = 6) associated with spasticity (P = 0.003; OR, 15.9) and ophthalmologic abnormalities (P = 0.016; OR, 16.9) |
Matos et al. 2021 [18] Cross-sectional case series study | • Cohort of 47 CZS newborns • Females (n = 24), males (n = 23) | Propose a classification scheme for orthopedic deformities and musculoskeletal complications by incidence, severity, and progression (non-spastic, spastic, arthrogrypotic) • Foot deformities: vertical talus, clubfoot (41.2%, n = 19) • Hip deformity (29.8%, n = 14); hip adduction contracture (20.2%), hip displacement (7.5%) • Knee deformity (12.8%, n = 6); knee displacement (6.4%) • Hypertonia (72.3%, n = 34) • Spasticity (77.3%, n = 36) • Arthrogryposis (15.2%, n = 7) associated with spasticity and more severe deformities • Hip and knee dislocation (29.8%, n = 14; 6.4%, n = 3) |
Contreras-Capetillo et al. 2021 [19] Case report | • Stillborn 35-week gestation • Sex unknown | • Zika viral RNA found in CSF • Craniofacial disproportion • Microcephaly • Irregular anterior and lower posterior hairline • Internal rotation and limited abduction of the shoulder • Limited hip abduction, knee extension, and flexion of both feet • Arthrogryposis • Absence of distal part of the right arm |
Tavares et al. 2021 [20] Cross-sectional case series study | • Cohort of 96 CZS cases • Age range 25 months to 40 months • Females (n = 41), males (n = 55) | • Positive pull to sit maneuver (45.8%, n = 44) • Positive in ventral suspension (32.3%, n = 31) • Positive in shoulder suspension (30.2%, n = 29) • Positive in scarf sign (32.3%, n = 31) • Hypertonia of appendicular muscles (94.8%, n = 91) • Quadriparesis (91.7%, n = 88), diparesis (3.1%, n = 3), hemiparesis (1.0%, n = 1) • Severe motor impairments, could be result of brain damage |
van der Linden et al. 2020 [21] Cross-sectional prospective study | • Cohort of 21 CZS cases • Age range 16 months to 30 months • Females (n = 14), males (n = 7) | Developmental delays in: • Total head control (14.3%, n = 3), partial head control (47.6%, n = 10) • Moro reflex (47.6%, n = 10), asymmetric tonic neck response (71.4%, n = 15), palmomandibular reflex (66.6%, n = 14), plantar grasp response (52.0%, n = 11), palmar grasp (76.2%, n = 16) • “125” posture of fingers (81.0%, n = 17), oromandibular dystonia (42.9%, n = 9), extensor axial hypertonia (38.1%, n = 8), “Swan neck” posture of fingers (18.8, n = 3), internal rotation of the shoulder (9.5%, n = 2) • Arthrogryposis (19.0%, n = 4) |
OR: odds ratio
Ophthalmic manifestations in CZS
| Study | Demographics | Findings |
|---|---|---|
Costa and Freitas 2022 [22] Cross-sectional case series study | • Cohort of 56 infants with CZS and microcephaly • Mean age 5.25 months ± 3.65 months • Females (n = 27), males (n = 29) | • Affected retina (79.2%, n = 19) and affected optic nerve (66.7%, n = 16) • Gross retinal pigmentation (45.8%, n = 11) • Macular chorioretinal atrophy/scarring (45.8%, n = 11) • Optic nerve hypoplasia (4.2%, n = 1) • Optic nerve increased cup-to-disk ratio (8.3%, n = 2) • Optic nerve pallor (58.3%, n = 14) |
Ventura et al. 2017 [23] Cross-sectional case series study | • Cohort of 32 infants with CZS • Age range 4 months to 7 months • Females (n = 15), males (n = 18) | • Microcephaly (n = 29) • Exotropia (34.4%, n = 11) • Esotropia (31.3%, n = 10) • Strabismus (9.4%, n = 3) • Nystagmus (28.1%, n = 9) • Astigmatism (45.3%, 29 of 64 eyes), Hyperopia (40.6%, 26 of 64 eyes), myopia (7.8%, 5 of 64 eyes) • Optic nerve hypoplasia (7.8%, 5 of 64 eyes) • Pallor (7.8%, 5 of 64 eyes) • Chorioretinal scar (17.2%, 11 of 64 eyes) • Pigment mottling (28.1%, n = 9) • Bilateral hypopigmented lesion (3.1%, n = 1) • Retinal vessel attenuation (3.1%, 2 eyes) |
de Paula Freitas et al. 2017 [24] Cross-sectional case series study | • Cohort of 29 infants with CZS • Age range 1 month to 6 months • Females (n = 18), males (n = 11) | • Microcephaly (34.5%, n = 10) • Ocular abnormalities (n = 10) • Ophthalmoscopic abnormalities (17 of 20 eyes in 10 children) • Ocular lesions (70%, n = 7) • Chorioretinal atrophy (64.7%, 11 of 17 eyes) • Severe chorioretinal atrophy in the macula (3 of 17 eyes), nasal retina (3 of 17 eyes), paramacular area (5 of 17 eyes) • Optic nerve abnormalities (47.1%, 8 of 17 eyes) • Bilateral iris coloboma (11.8%, 2 eyes of the same patient) • Lens subluxation (5.9%, 1 of 17 eyes) |
Ventura et al. 2016 [25] Case series | • 3 neonates born at 37 weeks to 38 weeks gestation with microcephaly and cerebral calcifications, presumed intrauterine Zika infection • Females (n = 2), males (n = 1) | • Gross macular pigment mottling (n = 3) • Foveal reflex loss (n = 3) • Macular neuroretinal atrophy (n = 1) • Fundoscopic alterations in the macular region (n = 3) |
Auditory manifestations in CZS
| Study | Demographics | Findings |
|---|---|---|
Muniz et al. 2022 [26] Prospective cohort study | • Cohort of 107 CZS cases, tested positive for Zika virus infection • Age range 0–3 years; 3-year follow-up auditory assessments • Females (n = 52), males (n = 53), (2 infants sex unknown) | • Hearing impairments in one or both ears (20.6%, n = 22) • 1:1 ratio of sensorineural: conductive hearing loss at last exam • No progressive hearing loss |
Craniofacial and oral manifestations in CZS
| Study | Demographics | Findings |
|---|---|---|
Díaz et al. 2023 [27] Cross-sectional case-control study | • Cohort of 14 CZS cases • Matched 12 healthy controls • Age range from 3 years to 5 years • Females (n = 9), males (n = 5) | • Microcephaly (92.9%, n = 13) • Dolichocephaly (71.4%, n = 10) • Mesocephalic (21.4%, n = 3) • Protruding maxilla (78.6%, n = 11) • Retrognathic mandible (64.3%, n = 9) • Asymmetric pupillary line (64.3%, n = 9) • Open bite (21.4%, n = 3) • Bruxism (85.7%, n = 12) • CZS microcephaly, maxillary prognathism, altered facial thirds, asymmetric pupillary line, bruxism (P = 0.006), deep and anterior open bite, and distal step decidual molar relationship (P = 0.031) |
da Costa et al. 2022 [28] Cross-sectional case series study | • Cohort of 10 children with CZS • Age range from 3 years to 5 years • Females (n = 3), males (n = 7) | • Microcephaly (90%, n = 9) • Cerebral palsy (100%, n = 10) • Oligodontia (20%, n = 2) • 3-year-old male had 12 primary dental absences, was microcephalic, had cerebral palsy, visual impairment, seizures, and hearing loss • 5-year-old male had 15 primary dental absences, had loss of bone density in maxilla and mandible, cerebral palsy, foot syndactyly, and ventriculomegaly |
Gomes et al. 2022 [29] Cross-sectional case-control study | • 62 cases of microcephaly CZS (n = 49), cytomegalovirus (CMV) (n = 4), toxoplasmosis (n = 1), other congenital infections (n = 8) • Age range from 7 months to 35 months • Females (n = 30), males (n = 32) | • Delayed tooth eruption (94%, n = 46) in CZS • Altered sequence of tooth eruption (72%, n = 33) in CZS • No statistically significant difference between CZS and other congenital infections with regards to delayed tooth eruption, alterations in sequence of tooth eruption of primary teeth, or developmental defects of enamel (opacity and hypoplasia) |
de Oliveira et al. 2020 [30] Cross-sectional case-control study | • Cohort of 45 CZS cases • 50 healthy controls • Mean age of 17 months • Females (n = 24), males (n = 21) | CZS is more likely to have: • Low weight • Nonexclusive breastfeeding (provision of food in addition to breast milk before age 6 months) • Mouth breathing • Swallowing difficulty • Excessive salivation • Hypoplasia of the middle third of the face (17.8%, n = 8) • Short lingual frenulum (2.2%, n = 1) • Abnormal insertion of upper labial frenulum (42.2%, n = 19) • Ogival palate (44.4%, n = 20) • Developmental defects of enamel (35.6%, n = 16) • Delayed tooth eruption delay (17.8%, n = 8) |
Carvalho et al. 2019 [31] Cross-sectional case-control study | • Cohort of 30 children with CZS and microcephaly evaluated over 24 consecutive months • 30 healthy controls • Females (n = 14), males (n = 16) • At the end of the study, 18 were younger than 25 months and 12 children were 25 months old or older • Delayed first dental eruption is considered after 9 months of age | • Delayed eruption of first tooth until after 9 months of age (60%, n = 18, 26.6 times more prevalent than in controls, P < 0.001, mean age of first dental eruption 10.8 months ± 3.8 months) • Alveolar ridge hyperplasia (3.3%, n = 1) • Short labial or lingual frenum (60%, n = 18, 4.9 times more prevalent than in controls, P = 0.028) • Inadequate lingual posture at rest (30%, n = 9, 26.9 times) • Micrognathia (6.7%, n = 2, P = 0.002) • Narrow palatine vaults (33.3%, n = 10, 24.8 times more prevalent than in controls, P = 0.01) |
This systematic review evaluated the clinicopathologic manifestations of CZS in children who were exposed in-utero to the Zika virus during the 2015–2016 Brazilian Zika outbreak. The eligible studies included 4 prospective cohort studies, 1 cross-sectional prospective study, 1 retrospective cohort study, 6 cross-sectional case-control studies, 8 cross-sectional case series, 1 small case series, and 5 case reports.
The combined number of CZS subjects examined in all of the studies was 856. However, it is assumed that some of the same patients had been recruited into more than one of the studies. Only two studies had over one hundred participants, Cavalcante et al. 2021 [10] and Muniz et al. 2022 [26]. Selection bias was inherent to the design of many of the studies to facilitate interrogation of specific associations amongst the more than seventy-six clinicopathological entities described to date and obstetric risk factors. Indeed, rather than pursuing epidemiological figures like incidence rates and prevalence metrics, several studies have successfully outlined proposals for developing classification schemes based either on the severity of symptoms (non-spastic vs. spastic arthrogryposis [18]), or the complexity of neuroradiologic findings [8].
Nevertheless, the numbers point to the CNS as a dominant target in CZS with severe consequences to the development of the head including the brain, the skull, and the face. Unfortunately, microcephaly appears to be inevitable, since babies born without it seem to develop it postnatally, and the outcomes do not seem to vary substantially for children born with or without microcephaly [10]. The occurrence of neuropathologic lesions is very notable with widespread cerebral calcifications (94%), quadriparesis (92%), epilepsy (91%), ventriculomegaly (89%), reduced cerebral parenchyma (86%), and malformation of cortical development (78%) and lack of gyri. Some of the oro-craniofacial deformities affect the mid-face more prominently and include a protruding maxilla (79%), with grinding teeth, short labial and lingual frenum (60%), and a higher prevalence of delayed chronology and altered sequence of primary dental eruption (P < 0.001) [29–31].
On the other hand, the auditory impairments affecting approximately 20% of infants have been shown to remain stable after birth [26]. The profound neurological dysfunction is then implicated in the development of cerebral palsy with a variety of muscle physiology impairments resulting in marked hypertonia (up to 92%), hypotonia (16%), spasms and spasticity (97%) which together with the skeletal defects in the feet (32%), hips (30%), knees (13%), and shoulders lead to the most severe outcome of arthrogryposis (15%) [18].
Immune dysregulation has emerged as a new field of interest with the discovery of system-wide deviations from normal values ranging from organ enlargement of the thymus, spleen, and lymph nodes, which was found in up to 81% of cases, to cell count increases in leukocytes (P < 0.005), hyper-segmented neutrophils, and atypical lymphocytes, and cytokine dysregulation with elevated levels of IFN-γ, IL-2, IL-4, and IL-5, and decreased levels of IL-1α, IL-7, IP-10/CXCL10, and G-CSF, which suggest impairment of immune functions at multiple levels [6].
The visual disturbances attributed to optic nerve (67%) and retinal (79%) lesions are currently considered to be complications, however, further scientific investigations on the teratogenic mechanisms of the Zika virus may reveal otherwise.
Discussion
In this paper, the many birth defects constituting the syndrome affecting the population of children who were born to mothers infected with Zika virus between 2015 and 2016 are compiled and analyzed. The review uncovered welcome research trends to conduct larger-scale studies for periods of years and frequent follow-up intervals with the purpose of exploring disease progression and formulating staging systems for the various clinical manifestations [8, 10, 18, 26].
CZS is characterized by microcephaly, microcalcifications, agyria, pachygyria, lissencephaly, ventriculomegaly, and other neurological disorders [6–15]. Moderate to severe intellectual disabilities and hearing loss are impacting the lives of affected children and their caregivers. In CZS, there is emerging evidence of immuno-suppressive alterations in the primary and secondary lymphoid organs [6]. Therefore, affected pediatric populations may be susceptible to recurrent infections and some have succumbed to pneumonia. The musculoskeletal manifestations that develop have been shown to worsen over time leading to joint dislocations, mobility limitations, falls, delayed speech, and motor developmental milestones [16–21]. Chorioretinal scarring and optic nerve lesions are complications that potentially lead to partial or complete loss of vision in CZS [22–25]. Jaw misalignment, weak muscles, and missing teeth can interfere with breast feeding, diet, and nutrition [27–31] regardless of etiology [29, 32]. Those who also have been affected by hearing loss may become deaf [26]. These abnormalities have been found to reduce the life expectancy of children born with CZS. According to Paixao et al. 2022 [33], the overall mortality rate was 11.3 times higher in children up to 36 months of age with CZS as compared to children of the same age born without the disease [95% confidence interval (CI)]. A multidisciplinary collaborative approach is crucial for the provision of nutritional, physical, psychological, educational, social, and healthcare support as they age into their preteen years.
Mechanisms of Zika virus infection on developing fetus
The complex molecular mechanism mediating the teratogenic effects of Zika virus infections is under investigation. After crossing the placental barrier, Zika virus would initially infect the placental trophoblasts and Hofbauer cells. Eventually, it must successfully cross the blood-brain barrier to infect fetal CNS progenitor cells. Indeed, studies have demonstrated alterations in tight junction proteins in association with Zika virus infection of brain vascular endothelial cells followed by infection of brain cells within the ventricular and subventricular zones, reviewed in [34].
Molecular pathways implicated in the pathogenesis of CZS involve Zika virus entry by binding to the receptor protein-tyrosine kinase (AXL) through growth arrest specific 6 (Gas6), endocytosis by clathrin, translocation, and activation of innate immunity pattern recognition receptors (PRRs) including Toll-like receptor 3 (TLR3), the retinoic acid-inducible gene I (RIG-I) DDX58, interferon induced with helicase C domain 1 (IFIH1), and other sensors of viral RNA [34].
Dysregulation of neuroinflammatory responses may facilitate Zika infections of radial glia, neural progenitor cells, microglia, and astrocytes. Microglial activation can perpetuate proinflammatory cytokine patterns that may further interfere with fetal neural tissue development, leading to defects in neural cell migration with consequent disorganization of cortical layers and abnormal brain structure. Microcephaly is associated with presence of infected neural progenitor cells, while brain calcifications correlate with increased viral load in cortical regions and infection of astrocytes [34].
The molecular mechanisms underlying the musculoskeletal abnormalities in CZS need to be further elucidated by investigating mutations in beta-tropomyosin, type 2 troponin, myosin heavy chain 3, and myosin binding protein 1 that might be associated with arthrogryposis [34]. Furthermore, brain defects in CSZ are likely to impair neuromuscular signaling and reduce intrauterine mobility, which would interfere with musculoskeletal development [35].
Similarly, the retinal and optic nerve lesions that develop in CZS may be consequences of neural progenitor cell disruptions or structural brain damage resulting in the progressive ophthalmologic complications [36]. Furthermore, the mechanism for hearing loss found in children is hypothesized to be a direct invasion of Zika virus into the neurons of the auditory pathway. It has been found that undifferentiated neurons are preferred targets resulting in congenital complications [37].
In addition, abnormalities in collagen-encoding genes such as collagen type I alpha 1 chain (COL1A1), collagen type II alpha 1 chain (COL2A2), peptidylprolyl isomerase B (PPIB), osteoglycin (OGN), and serpin family H member 1 (SERPINH1) may be responsible for the craniofacial defects seen in CZS. These genes play a crucial role in the formation of collagen fibrils, osteoblast differentiation, and bone development [38]. Infection of the human neural progenitor and neural crest cells by the Zika virus is also believed to cause the failure of dental eruption [31, 39]. Based on these findings, it can be concluded that by crossing the placental barrier and invading fetal progenitor cells, Zika virus infection triggers a cascading series of teratogenic effects that warrant further investigation.
In conclusion, this systematic review demonstrates that CZS has many drastic effects on children born to infected mothers including microcephaly, brain calcifications, restricted joint mobility, retinal scarring, hearing loss, immune system alterations, and delayed teeth eruption. The mortality rates in neonates and infants born with CZS are higher than in those without the disease. The molecular mechanism for Zika infections is not entirely clear but it is hypothesized that the virus crosses the blood-brain barrier in a fetus and causes multiple defects in development. Based on these findings, further studies should explore (1) the longitudinal monitoring of motor development through systematic assessments, (2) new public policies that promote supportive and inclusive environments for children with CZS, (3) building relationships between health, education, and social service actions for implementation and management of comprehensive health care for this vulnerable population of children, and (4) investigating the molecular mechanism of teratogenicity following transplacental Zika infection.
Abbreviations
| CDC: |
Center for Disease Control and Prevention |
| CNS: |
central nervous system |
| CSF: |
cerebrospinal fluid |
| CXCL10: |
C-X-C motif chemokine ligand 10 |
| CZS: |
congenital Zika syndrome |
| EEG: |
electroencephalogram |
| G-CSF: |
granulocyte-colony stimulating factor |
| IFN-γ: |
interferon-gamma |
| IL-2: |
interleukin-2 |
Declarations
Acknowledgments
We are grateful for the support of Dr. Branka Filipovic, head of research, at the Saint James School of Medicine, for her early input in designing this study. We thank Mr. Oscar Andrade, chief librarian, at the Saint James School of Medicine, as well as the Kean University Library Nancy Thompson Learning Commons for assistance with accessing the full-text, peer-reviewed articles that were needed to conduct this review.
Author contributions
Dhaara S and Dhairavi S: Conceptualization, Methodology, Investigation, Writing—original draft, Writing—review & editing. OM: Conceptualization, Methodology, Investigation, Writing—original draft. RZ: Conceptualization, Methodology, Investigation, Writing—review & editing.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publication
Not applicable.
Availability of data and materials
The data and materials obtained for this systematic review were obtained online using search engines and the research databases mentioned in the methodology. The articles referenced in this paper can be accessed freely by searching for them on PubMed, PMC, Ovid, Cochrane, or Google Scholar.
Funding
This research was supported by Kean University Office of Research and Sponsored Programs (ORSP). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Copyright
© The Authors 2024.