Affiliation:
1Foodomics Laboratory, Institute of Food Science Research (CIAL), CSIC, 28049 Madrid, Spain
ORCID: https://orcid.org/0000-0003-2127-8303
Affiliation:
2Dipartimento di Scienza e Tecnologia del Farmaco, Università Di Torino, 10125 Turin, Italy
ORCID: https://orcid.org/0000-0003-1782-7357
Affiliation:
3Department of Agricultural and Food Sciences, Alma Mater Studiorum Università di Bologna, 40126 Cesena, Italy
ORCID: https://orcid.org/0000-0002-5543-2359
Affiliation:
4Key Laboratory of Analytical Chemistry for Living Biosystems, Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, China
5Huiyin Institute of Technology, Huaian 223003, Jiangsu, China
ORCID: https://orcid.org/0000-0003-4426-406X
Affiliation:
7Institute of Physical and Analytical Chemistry, School of Exact and Natural Sciences, Tbilisi State University, Tbilisi 0179, Georgia
Affiliation:
8Chemical and Food Engineering Department, Federal University of Santa Catarina, Florianópolis 88040-970, Brazil
ORCID: https://orcid.org/0000-0003-3393-0995
Affiliation:
9Hamburg School of Food Science, Institute of Food Chemistry, University of Hamburg, 20146 Hamburg, Germany
Affiliation:
10Department of Food Science, National Pingtung University of Science and Technology
ORCID: https://orcid.org/0000-0002-4904-0519
Affiliation:
11Packaging Group, Institute of Agrochemistry and Food Technology (IATA-CSIC), 46980 Paterna, Spain
ORCID: https://orcid.org/0000-0001-6376-8939
Affiliation:
1Foodomics Laboratory, Institute of Food Science Research (CIAL), CSIC, 28049 Madrid, Spain
ORCID: https://orcid.org/0000-0002-7214-6653
Affiliation:
12Department of Medicine, Democritus University of Thrace, 68100 Alexandroupolis, Greece
ORCID: https://orcid.org/0000-0001-7835-8246
Affiliation:
13School of Environmental Science and Engineering, Tianjin University, Tianjin 300354, China
ORCID: https://orcid.org/0000-0001-5496-3011
Affiliation:
14Department of Chemistry and Technology of Drugs, Sapienza University of Rome, 00185 Rome, Italy
ORCID: https://orcid.org/0000-0001-8659-5890
Affiliation:
15DNA & RNA Sensing Lab, Universidade de Trás-os-Montes e Alto Douro, Departamento de Genética e Biotecnologia, Blocos Laboratoriais Ed, 5000-801 Vila Real, Portugal
16Universidade de Lisboa, Faculdade de Ciências, BioISI – Biosystems & Integrative Sciences Institute, 1749-016 Lisboa, Portugal
ORCID: https://orcid.org/0000-0001-8229-0117
Affiliation:
1Foodomics Laboratory, Institute of Food Science Research (CIAL), CSIC, 28049 Madrid, Spain
ORCID: https://orcid.org/0000-0001-6709-349X
Affiliation:
17National and Kapodistrian University of Athens, Department of Chemistry, Food Chemistry Laboratory, Panepistimiopolis Zografou, 15771 Athens, Greece
Affiliation:
18Environmental and Computational Chemistry Group, School of Pharmaceutical Sciences, Zaragocilla Campus, University of Cartagena, Cartagena 130014, Colombia
ORCID: https://orcid.org/0000-0003-3089-5872
Affiliation:
19Environmental and Food Safety Research Group of the University of Valencia (SAMA-UV), Desertification Research Centre CIDE (CSIC-UV-GV), 46113 Valencia, Spain
Affiliation:
17National and Kapodistrian University of Athens, Department of Chemistry, Food Chemistry Laboratory, Panepistimiopolis Zografou, 15771 Athens, Greece
ORCID: https://orcid.org/0000-0002-3450-5969
Affiliation:
20Department of Food BioSciences, Teagasc Food Research Centre Ashtown, D15 DY05 Dublin, Ireland
ORCID: https://orcid.org/0000-0002-8073-4981
Affiliation:
21Department of Pharmacy, University of Salerno, 84084 Fisciano, Italy
22National Biodiversity Future Center (NBFC), 90133 Palermo, Italy
ORCID: https://orcid.org/0000-0003-0718-5450
Affiliation:
23Regional Institute of Applied Scientific Research, University of Castilla – La Mancha, 13004 Ciudad Real, Spain
24Department of Analytical Chemistry and Food Technology, Faculty of Chemical Science and Technology, University of Castilla – La Mancha, 13071 Ciudad Real, Spain
ORCID: https://orcid.org/0000-0003-1728-3097
Affiliation:
25Food Quality and Design, Wageningen University & Research, 6708 Wageningen, The Netherlands
26Division of Human Nutrition and Health, Wageningen University & Research, 6708 Wageningen, The Netherlands
ORCID: https://orcid.org/0000-0002-1634-2334
Affiliation:
27University of Coimbra, Faculty of Pharmacy, 3000-548 Coimbra, Portugal
28Centre for Animal Science Studies (CECA), ICETA, 4099-002 Porto, Portugal
29Associate Laboratory for Animal and Veterinary Sciences (Al4AnimalS), 1300-477 Lisbon, Portugal
ORCID: https://orcid.org/0000-0002-0226-921X
Affiliation:
30Grupo de Investigación en Polifenoles (GIP-USAL), Facultad de Farmacia, Universidad de Salamanca, Campus Miguel de Unamuno, 37007 Salamanca, Spain
ORCID: https://orcid.org/0000-0001-6592-5299
Affiliation:
31Department of Medicine, College of Medicine, Korea University, Seoul 02841, Republic of Korea
32Centro de Estudios Tecnológicos y Universitarios del Golfo, Centro, Veracruz 91700, Mexico
ORCID: https://orcid.org/0000-0002-7301-8151
Affiliation:
33Departamento de Química, Faculdade de Ciências Exatas e da Engenharia da Universidade da Madeira, Campus Universitário da Penteada, 9020-105 Funchal, Portugal
34CQM - Centro de Química da Madeira, NPRG, Universidade da Madeira, 9020-105 Funchal, Portugal
ORCID: https://orcid.org/0000-0003-1965-3151
Affiliation:
35CBQF - Centro de Biotecnologia e Química Fina – Laboratório Associado, Escola Superior de Biotecnologia, Universidade Católica Portuguesa, 4169-005 Porto, Portugal
ORCID: https://orcid.org/0000-0001-8912-9836
Affiliation:
36Oil Crops Research Institute of Chinese Academy of Agricultural Sciences, Key Laboratory of Biology and Genetic Improvement of Oil Crops, Key Laboratory of Detection for Mycotoxins, National Reference Laboratory for Agricultural Testing (Biotoxin), Hubei Hongshan Lab, Wuhan 430062, Hubei, China
Affiliation:
1Foodomics Laboratory, Institute of Food Science Research (CIAL), CSIC, 28049 Madrid, Spain
Email: a.cifuentes@csic.es
ORCID: https://orcid.org/0000-0002-7464-0217
Explor Foods Foodomics. 2024;2:707–766 DOI: https://doi.org/10.37349/eff.2024.00060
Received: September 19, 2024 Accepted: November 12, 2024 Published: November 28, 2024
Academic Editor: Cem Erkmen, Istanbul Aydin University, Türkiye
In this perspective article, several internationally recognized experts, members of the editorial team of this journal, discuss a selection of current hot topics identified in Food Science and Foodomics. The topics are comprised of the main areas of Food Science and Foodomics, namely, food safety, food authenticity, food processing, and food bioactivity. Logically, several of the discussed topics involve more than one of the mentioned main areas. Regarding food safety, the topics discussed are the use of analytical nanotechnology, nanometrology, nano-chromatography; the determination of organic contaminants based on MS and NMR; the impact of microplastics and nanoplastics on food or the contamination of foods with plant toxins. Regarding food authenticity, the paper discusses the role of MS, NMR, biosensors and the new trends in foodomics for food authentication. In terms of food processing, the work shows interesting perspectives on novel processing technologies, the effect of food processing on the gut microbiota or in the interaction among secondary metabolites and macromolecules; the development of active packaging, and the potential effects of introducing recycled plastics in food packaging; the new green extraction and encapsulation strategies of bioactive compounds from food by-products; and the anti-biofilm capacity of natural compounds/extracts/vegetal oils and essential oils. Food bioactivity and the relation between food and health includes the bioavailability and bioaccessibility of bioactive compounds; new trends and challenges in the interaction of nutraceuticals with biological systems; how food matrix impacts the bioaccessibility of nutrients and bioactive compounds; or the study of biodiversity, food and human health through one-health concept. We anticipate elaborations on these hot topics will promote further studies in Food Science and Foodomics.
Today, the challenges facing Food Science and Foodomics are enormous in both, number and complexity. This is well in line with the current speed of evolution of the Global Market and how our knowledge about many different issues that will impact our future has increased and deepened. Thus, these important issues begin with the way we produce our food, including its sustainability, the interaction with our planet in terms of use or reduction of pesticides, herbicides, etc., and the preservation of biodiversity along with the study of new alternative species; the identification and investigation of emerging risks in terms of food safety, food authenticity and food processing, as well as possible criminally motivated manipulations, which can be summarized under the term food crime. Innovative analytical approaches address and overcome these novel risks; the development of new food processing and technological innovations and their effects on food quality; and finally, the in-depth study of the positive or negative health effects of foods and the understanding of the mechanisms that may explain these effects. All of these topics should be included in the framework of a necessary Circular Economy considering how the food production system as a whole affects society and sustainability. Although the study of many of these issues is still in its infancy, luckily, we count now with powerful analytical techniques and robust technologies that can help us determine and, in several cases, demonstrate some of these issues. Below is a selection of some of the hot topics identified by the Editorial Team of Exploration in Food Science and Foodomics (see Figure 1). We believe these topics will attract a lot of attention from the researchers working in this important field.
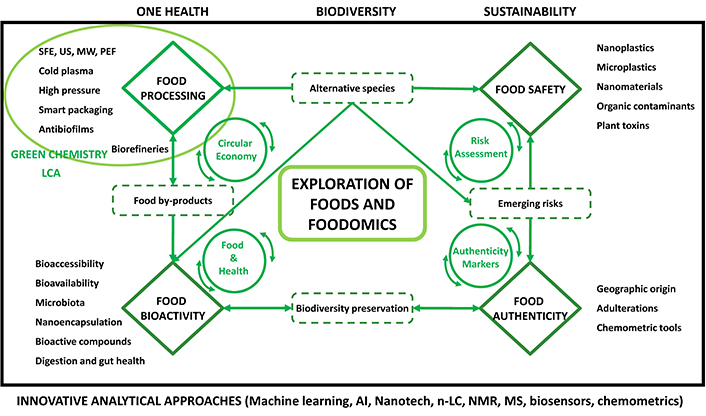
Framework defining the future trends in Food Science and Foodomics identified by the Editorial Board of EFF (Exploration in Food Science and Foodomics, Source: the authors). MW: microwave; PEF: pulsed electric field; SFE: supercritical fluid extraction; US: ultrasound
Nanoscience and nanotechnology have emerged in recent years as a revolutionary tool in almost all fields of science, including food science. Nanomaterials (NMs), according to the European Union (EU) definition, are natural, incidental or manufactured material containing particles, in an unbound state or as an aggregate or as an agglomerate and where, for 50% or more of the particles in the number size distribution, one or more external dimensions is in the size range 1–100 nm [1]. NMs are present ubiquitously around us and they can pose risks to human or animal health, or harmful environmental effects. NMs are commonly used in food industry as food additives, food ingredients, and also as food contact materials. Some references illustrate the potential toxicity of NMs in humans [2, 3], and more recently the European Food Safety Authority (EFSA) Scientific Committee has reported a guidance of risk assessment of NMs to be applied in food and feed chain [4].
It is recognized that food ingredients that are generally considered as safe at the macro level may be not safe at the nanoscale. NMs, in particular can pass through cells and interfere with several subcellular mechanisms. The NMs can even penetrate into cell nuclei and affect the DNA, as it is reported in some references [5, 6]. Therefore, the detection of NMs in food products is essential. Analytical nanometrology (ANM, or metrology applied to NMs) deals with the application of the metrology at this size-level (nanoscale) [7].
Different analytical techniques, as outlined in Table 1 (including the abbreviation of each technique), are applied in the characterization of NMs in food to determine their size distribution, composition, structure, shape, surface, level of agglomeration, and concentration. The complete characterization is commonly impossible, but the studies must be addressed to those critical features depending on the intendent use. Microscopic techniques, such as SEM, TEM and STEM, give information about the size (size distribution), morphology, shape, and level of agglomeration; diffraction techniques (XRD, SERS mainly), and spectroscopic techniques (FTIR mainly) give information about the composition, structure and crystalline state. All these techniques need to be NMs (samples) in solid state, but in many cases NMs and samples are presented in solution (basically in aqueous solutions). In these cases, UV-vis and fluorescence spectroscopy can sometimes give information on size and concentration. Here, it is very important the role of DLS because inform about the size distribution in solution, it means the hydrodynamic size of NMs including the hydration sphere associated with the NM. Even more, information about the so-called zeta-potential. This is the voltage difference existing between the surface of the nanoparticle (NP) and the counter-ions strongly bound to this particle when it is moving in an electric field. NMs characterization is an important issue when they are going to be used for other applications or purposes (as it may be in food field as additives, preservatives, stabilizers, etc.), but not for nanometrological purposes such as identification and determination of NMs in specific food samples [7]. Here, the role of instrumental separation techniques and some spectrometric techniques are critical tools. Within instrumental separation techniques, hydrodynamic chromatography (HDC), SEC, CE, and more recently and important, field flow fractionation techniques (FFF) have been employed, especially when these techniques are hyphenated to the wide variety of mass spectrometer detectors. Today, ICP-MS has emerged as a definitive technique for NMs detection/determination, with many advantageous possibilities, particularly in the “single-particle” mode (sp-ICP-MS). The sp-ICP-MS allows to measure individual inorganic NPs, with differentiation between ionic and particulate signal without any prior separation, which is very sensitive and permits to give element specific information including particle concentration and size distribution. Some examples in food field of the use of sp-ICP-MS can be illustrative [8, 9], and a general overview was recently reported by Villamayor et al. [10].
Some selected examples of the analytical characterization and determination of NMs in the food field according to the technique involved
| Type of information provided | More common analytical techniques (*) | Selected applications (references) |
|---|---|---|
| Size distribution, shape, aggregation state, surface | SEM, TEM, AFM, STEM, DLS, ICP-MS, HPLC, CE, FFF, SEC | [11–14] |
| Hydrodynamic diameter, surface charge, zeta potential | DLS | [11, 12, 14] |
| Structural characterization | RS, LIF, UV-vis, IR, FTIR, XRF, XPS, NMR, MS | [15–22] |
| Quantification | ICP-MS, sp-ICP-MS, SEC, HDC, FFF | [8, 23–28] |
(*) SEM: scanning electron microscopy; TEM: transmission electron microscopy; AFM: atomic force microscopy; STEM: scanning TEM; DLS: dynamic light scattering; ICP-MS: inductively coupled plasma-mass spectrometry; HPLC: high performance liquid chromatography; CE: capillary electrophoresis; FFF: field flow fractionation; SEC: size exclusion chromatography; RS: Raman spectroscopy; LIF: laser-induced fluorescence; UV-vis: ultraviolet-visible spectroscopy; IR: infrared spectroscopy; FTIR: Fourier transform infrared spectroscopy; XRD: X-ray diffraction; XRF: X-ray fluorescence; XPS: X-ray photoelectron spectroscopy; NMR: nuclear magnetic resonance; MS: mass spectrometry; sp-ICP-MS: single particle ICP-MS; HDC: hydrodynamic chromatography
In spite of the importance and rise of analysis of NMs in food, challenges exist for a comprehensive and complete analysis of samples containing NMs. Below are some of bottlenecks to end users (routine/control analytical laboratories):
Identification of real problems and situations where nanocomponents may be found in specific samples.
Sample treatment assuring the integrity of the nanocomponents.
Development of screening systems for a rapid response (e.g., the development of sensing platforms is a crucial need).
Need of reliable standards for key activities assuring the traceability and the reliability of the results for calibration and certified reference materials for method validation.
Need of quality assurance programs to harmonize analytical methodologies for NMs in specific samples.
Finally, it is important to note that the analysis of inorganic and organic NMs requires different analytical strategies. ANM for inorganic NMs is more advanced (e.g., ICP-MS and sp-ICP-MS, etc.) since more attention has been paid to this type of NMs. ANM for organic NMs (very important in the food field) needs further developments [probably with different modalities of liquid chromatography, CE and FFF in combination with mass spectrometry (MS) and MS/MS]. Thus, the term nanostructured organic materials (NOMs) have been used to include nanoemulsions (NEs), nanoliposomes (NLs), solid lipidic NPs (SLNs), and nanostructured lipidic carriers (NLCs), and here the analytical control of nanodelivery lipid-base systems for encapsulation of nutraceutical compounds presents some achievements, but many challenges today in the general framework of the ANM [28]. Examples such as curcumin/nanocurcumin [29], quercetin/encapsulated nano-quercetin [30], and nanoencapsulated vitamin D3 [31] are good proofs of the different analytical strategies with respect to those followed by inorganic NMs.
Analytical chemistry plays a crucial role in food science and food industry, involving both food quality and safety control. After integration with nanotechnology and nanoscience, this new branch, called analytical nanotechnology (ANT) was gradually established in the first decade of this century [32]. ANT is seemingly beginning to mature now as it is indicated by the significantly increased exploration of ANT methods and their application to food science and food industry.
Despite its complexity, food can now be analyzed using highly selective, fast and efficient analytical methods. However, there are still quite a few practical challenges that need to be addressed, such as how to improve further the analytical selectivity, separation efficiency, and detection sensitivity; lower the operation difficulty, technical barriers, and material and sample consumption; simplify sample pretreatment, accelerate analytical speed, and reduce analysis costs. Fortunately, as nanoscience and nanotechnology emerge, at least two novel strategies have been shown up in addressing those challenges. The first strategy is to develop ANT through the integration of nanoscience and nanotechnology with other scientific disciplines and technologies, such as electronics, chip technology, biology, biochemistry, and especially analytical chemistry; while the second one is through direct exploration of nano- (scientific and/or technological) principles. In practical research, the two strategies are rarely used separately, instead, they are often applied together or alternately. Various ANT methods have been established, for example, gold immunochromatography assay (GICA), nano-based colorimetric immunoassay, nano-amplified surface plasmon resonance (nSPR), optical nanosensors, quantum dot-based spectrometry, electrochemical NPs, NPs-mediated mass sensors (e.g., quartz crystal microbalance or QCM, and microcantilever), magnetic nanosensors, and so forth.
In the development of ANT methods, the top choice is to explore or utilize NMs, especially nanosensors, because they can have adjustable recognition elements or affinity ligands (e.g., enzymes, antigens or antibodies) for the specific recognition of target analytes. It is also critical that NPs and/or their nanostructure can exhibit unique target signals in highly complex food matrices [33, 34]. ANT methods can further improve their detection selectivity and/or sensitivity through functionalizing the NPs to increase their affinity and/or to add optical absorbance or pseudo mass to the target analytes. The functionalization of NMs is usually achieved by tagging or staining the analytes during either step of sample pretreatment, separation or detection. This is also applicable to the amplification of the transduced signals of nanosensors [35–37]. In addition, NMs also involve the innovative fabrication and application of food analysis-oriented nano-noses and nano-tongues [32].
Nanosensors are possibly the earliest version of ANT, and can be traced back to the colloidal gold staining technique reported in 1971 [38] or even earlier in 1939 when the fine particles of gold NPs-labeled tobacco mosaic virus were observed under an electron microscope [39]. After about half a century of gradual development and accumulation, ANT methods started to rapidly develop in the first decade of this century [32, 40–42]. It is now becoming one of the advanced methodologies in food science. The expected new development trend is to explore fast food assaying or screening methods, with a focus also on nanosensors that can be represented by NPs-based immunochromatography [43]. It is in this case that ANT may be narrowed to nanodetection or nanosensors that are often classified as biosensors when immunochemistry and/or biology are integrated.
ANT involves also simplifying the sample pretreatment. Automated or programmatic NPs-based sample pretreatments can avoid artificial and non-artificial errors in the fast capture, purification, cleanup and concentration of various nutrients or harmful substances (e.g., microbial genetic materials, proteins, toxins, allergens, processing-introduced deleterious chemicals, etc.). With dispersive affinity NPs, a target analyte can easily be captured and isolated from its complex food matrices simply through precipitation, centrifugation or magnetic attraction [44–48]. NMs can be used as novel absorbents and integrated into common sample pretreating techniques. NPs are advantageous over other adsorbents because they possess reactive surface that enables surface functionalization prior to or during sample treating process. NPs can also be used to fast clean up the food samples based on their extremely high specific surface area [49].
While traditional separation tools such as chromatography and electrophoresis have achieved significant advances in the analysis of various NMs [50, 51], they have evolved to capillaries and micro-chips sizes, and many packed media to sub-micrometer particles, which makes their consumption of solvents and samples down to nL or sub-nL scale. Furthermore, both bare and modified NPs can be integrated into or directly serve as high performance stationary phases and/or act as mobile phase additives in chromatography and electrophoresis [52–56]. Clearly, the research on ANT methodology is very diverse and although some of ANT kits and sensor devices are commercially available now, we still lack international standard evaluation methods.
ANT methods can broadly be applied to food science and food industry because they can analyze exogenous compounds (e.g., pesticides, toxic anions, ripening gases or vitamin supplements) and endogenous substances (from microorganisms to molecules such as vitamins) [57]. This can particularly be represented by the detection of various foodborne microorganisms with nanosensors, which is originated from the early electron microscopic observation of Vibrio cholera O1 [58]. The pathogenic bacteria in food already studied include Escherichia coli (e.g., O157:H7), Salmonella, Shigella, Vibrio cholerae, Vibrio parahaemolyticus, Proteus, Listeria monocytogenes, Listeria ivanovii, Bacillus cereus, Clostridium botulinum, and Campylobacter jejuni, etc. [59–68]. These microorganisms can either be detected directly through bacterial membrane receptors or indirectly through their generic materials such as specific DNA fragments, or even by their specific metabolites. In SPR or QCM of bacteria, NPs can be used as both capture probes and amplifiers to acquire high gain of signals [69] and in turn to shorten the detection time from days to minutes.
Nanosensors are also applicable to the fast (in minutes) assay of endogenous and exogenous molecules such as hormones [70], pesticides, insecticides and their residues [71–73]. This is exemplified by the analysis of mycotoxins. It is known that there are about 25% of crops per year contaminated with mycotoxins (more than 200 mycotoxins from about 150 fungal species). These toxic substances in foods and feeds can cause poisoning and diseases in humans [74, 75]. The highly toxic ones, such as botulinum toxin, ochratoxin and aflatoxin, have been extensively studied and can be detected at a concentration down to pg/mL level [76, 77] by GICA or its modified approaches [36, 71, 78–84], colorimetric immunoassay [85], nSPR [86–89], optical sensors [90–94] including especially quantum dots [95–102], electrochemical sensors [93, 103–105], NPs-mediated mass sensors [106, 107], and NPs-based magnetic sensors [107–109]. In addition, some novel NPs have also been explored and tried in ANT such as nano-metal-organic framework materials and other nanocomposites [110–114] for the detection of other substances [70, 115], such as oxygen, ethylene, biogenic amines, hypoxanthine, polyphenol, glucose, folic acid, melatonin, cyanide, etc. [32, 116].
In the analysis of large multi-analytes such as pesticides, nanoflow chromatography has the significant advantage when it is coupled with MS for identification and quantification of numerous trace peaks. For example, nanoflow liquid chromatography coupled to Q-Exactive Orbitrap with full-scan high resolution was assessed for over 60 representative pesticides in tomato, baby food, orange, fruit-based jam and olive oil samples [117]. The high sensitivity enables beyond 1:50 dilution factors in pesticide residue measurement, with limits of quantitation at low ng/kg. Matrix-matched standards may then be skipped, which simplifies laboratory workflows. Robust separation-based ANT methods will facilitate foodomics study [118].
In short, ANT as represented by versatile nanosensors [119] has undergone rapid development and shown attractive applications in food science and food industry. With various advantages (e.g., used individually or in an array form), inserted in different food processing steps and/or locations (including food microbial culture), ANT can easily adapt to different styles and/or states (gaseous, liquid, and solid; spoiled or fresh, etc.) of packaged or unpackaged foods to conduct qualitative or quantitative detection of various trace pathogens, organic compounds and other chemicals with high selectivity, responsiveness and recovery. ANT can be expected to develop even faster from now on. Nevertheless, more effort has to be paid to the exploration, simplification and evaluation of cost-effective methods. It is also crucial to promote methodological commercialization and to develop international standard evaluation methods.
Nano-liquid chromatography (nano-LC) is a microfluidic technique mainly used for analytical purposes. It has gained interest in the research and application fields because its features are alternative/complementary to HPLC. The compounds’ separation is performed in capillaries of low internal dimensions (10–100 μm I.D.) containing a selected stationary phase under a flow rate of 10–700 nL/min. The low flow rate 1) reduces the analytes dispersion offering high mass sensitivity, 2) makes the technique eco-friendly, and 3) allows a perfect coupling with MS. The stationary phases used in HPLC have been employed in nano-LC with its different modes, namely packed (mainly based on silica-modified particles) or in monolithic material, and bonded/adsorbed on the capillary wall [open tubular (OT)-nano-LC].
Nano-LC has been successfully applied for the separation and analysis of compounds studied in different areas, e.g., proteomics, pharmaceutical, agrochemical, environment, biomedicine, food chemistry, etc. The applicability of nano-LC in the field of separation science has been documented by some review articles where the main features and applications have been reported [118, 120–127].
Some examples document the practical applicability of nano-LC to food analysis. For instance, the analysis of some amino acids enantiomers has been performed after derivatization with fluorescein isothiocyanate (FITC) in fruit juice with OT-nano-LC where β-cyclodextrin and FITC were the chiral selectors [128], while D’Orazio et al. [129] analyzed amino acid enantiomers after derivatization with FITC in orange fruit juice using a capillary column packed with RP18 stationary phase. In both examples, an indirect method was applied. Amino acid enantiomers were also separated by nano-LC and capillary electrochromatography (CEC). Analytes were derivatized with FITC and separated in a capillary column containing silica particles with coated polysaccharides [cellulose tris(3-chloro-4-methylphenylcarbamate)]. The content of citrulline in a dietary supplement was measured by CEC [130]. The analysis of 3-mercaptohexan-1-ol, 3-mercaptohexylacetate, and their corresponding disulfides in wine was reported providing partial information on the wine aroma evolution [131]. Compounds, after derivatization with N-ethylmaleimide or N-phenylmaleimide were analyzed with a nano-LC-MS-MS in the presence of an RP18 stationary phase. Caffeine and riboflavin were separated and analyzed in dietary supplements by nano-LC using a column (150 mm × 100 μm I.D.) packed with Zorbax 300SB C18 (3.5 μm particle size) [132]. Another interesting application includes the analysis of oligosaccharides in goat colostrum employing a nano-LC-Chip–Q-TOF MS; the chip (analytical column) was packed with graphitized carbon [133]. Although the good results achieved by nano-LC and the features of this technique, some potential hurdles still need to be overcome, e.g., only a few types of capillary columns are commercially available. This problem can be resolved by preparing the columns in the own laboratory, however, it requires experience and ability.
With the continuous advancement of global industrialization and urbanization, plastic pollution has become an environmental issue that cannot be ignored [134, 135]. In particular, the widespread presence of microplastics and nanoplastics (MNPs) poses a serious threat to the environment and the health of living organisms. Due to their small particle size, MNPs can easily spread through environmental media and food chains, thereby potentially impacting the environment and human health [136]. These tiny plastic particles are now ubiquitous, having even invaded the human body. People are continuously exposed to these MNPs through food and beverage consumption, air inhalation, and cosmetics, which may pose potential health risks [137, 138]. Figure 2 shows an overview of sources of microplastics, pathways of degradation and human exposure routes and consequences, as described by Mamun et al., 2023 [137]. However, research on the impact of MNPs on food production and quality is still insufficient at present, lacking systematic analysis and evaluation. This has led to a limited understanding of the role and impact of these tiny plastic particles in food production and quality. Therefore, conducting relevant research is of great significance for formulating corresponding management and control measures, ensuring food safety, and promoting sustainable development.
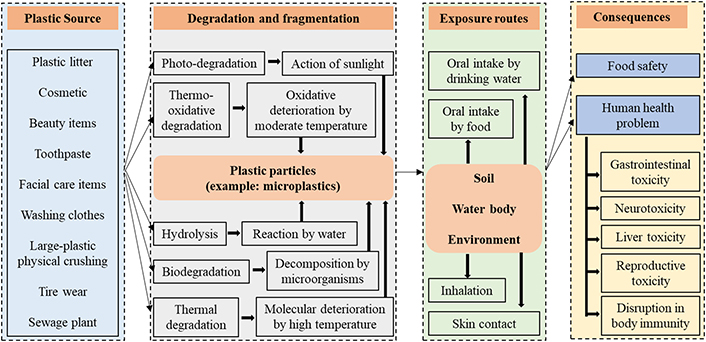
Overview of sources of microplastics, pathways of degradation and human exposure routes and consequences (reproduced with permission from Mamun et al. [137], Science of the Total Environment, 2023, 858, 159834; Figure 3; © 2022 Elsevier B.V.)
Microplastics are plastic fragments, particles, or fibers smaller than 5 mm in size, originating from the breakdown, abrasion, or manufacturing residues of larger plastic items. Nanoplastics, on the other hand, are plastic particles or fibers sized at the nanometer scale (1 to 100 nm). The main sources of MNPs include plastic waste, tire wear, and textile washing. The formation of MNPs occurs mainly through physical, chemical, and biological processes [135]. For example, plastic waste, such as discarded plastic bags, bottles, and packaging, undergoes long-term weathering and degradation processes in the natural environment, gradually forming tiny plastic particles.
MNPs infiltrate the soil through various means such as agricultural activities and wastewater discharge [139]. Their presence not only disrupts the natural structure of the soil, affecting its aeration and water retention, but also has long-term impacts on soil ecological balance. Specifically, these plastic particles can clog soil pores, reducing soil permeability, which in turn affects the respiration and water absorption of plant roots. In addition, the intrusion of MNPs may lead to fundamental changes in soil microbial communities [136, 140]. Soil microorganisms are crucial for maintaining soil fertility and ecological balance, with their diversity and function directly affecting the healthy growth of crops. These tiny plastic particles can interact with microorganisms, disrupting the original microbial community structure and potentially reducing or eliminating certain beneficial microorganisms. This imbalance in microbial communities can further impact soil fertility, negatively affecting crop growth and yield. Furthermore, MNPs may also act as carriers of toxic substances in the environment. Due to their special surface properties, they can easily adsorb harmful substances such as heavy metals and pesticides from the environment [141–143]. Over time, these toxic substances can accumulate in the soil and enter organisms, including humans, through the food chain, posing potential health risks.
MNPs can pollute drinking water resources through rivers, lakes, and other water bodies. These tiny plastic particles are difficult to be completely removed through conventional water purification processes [144]. MNPs can cause direct harm to aquatic organisms, such as ingestion and physical damage, affecting their survival and reproduction [145, 146]. This damage can lead to a reduction in the number of aquatic organisms and a decrease in biodiversity, causing long-term impacts on the balance of aquatic ecosystems. This not only threatens the survival of aquatic life but also exacerbates water resource pollution, creating a vicious cycle. In agriculture, irrigation water containing MNPs poses a potential threat to crop growth and quality [147]. These small particles may adhere to crop surfaces, affecting photosynthesis and respiration, thereby reducing crop yield and quality. These plastic particles can also be absorbed by crops, entering their internal tissues and posing potential health risks to humans.
During the crop’s growth, the presence of MNPs has become a potential threat. They can enter the crop growth environment through soil and water sources, affecting crops in various ways. Firstly, MNPs can directly hinder crop growth and development, leading to phenomena such as slow growth and yellowing leaves. This not only affects the appearance of crops but can also reduce crop yield and quality, causing direct economic losses to agricultural production [148]. MNPs can block crop roots, which are vital for water and nutrient absorption. Once blocked, crop roots cannot function properly, leading to nutrient deficiencies in crops [149]. This nutrient deficiency can affect normal crop growth, further impacting crop yield and quality, and causing long-term effects on agricultural production. Secondly, MNPs can adsorb toxic substances from the environment and accumulate within crops [142]. These toxic substances can enter the human body through the food chain, posing potential health risks.
MNPs have high environmental mobility and bioaccumulation potential due to their small size. They can contaminate food raw materials through air and water sources [139]. These particles can adhere to the surfaces of agricultural products or mix into water sources used for irrigation or food processing, entering the food chain [150–152]. Their presence can affect the taste and appearance of food and, more importantly, may carry harmful substances, posing potential health risks to humans [153]. Contamination during processing is also a concern. During food processing, plastic equipment, containers, and packaging materials may come into direct contact with food, providing a pathway for MNPs contamination.
Additives in plastics, such as plasticizers, stabilizers, and pigments, can migrate during plastics contact with food. The migration of these additives can alter the chemical composition of food, posing potential health threats to humans. Phthalates, commonly used as plasticizers in plastics, have potential endocrine-disrupting effects. Some plastics may contain persistent organic pollutants (POPs), which are difficult to degrade in the environment and can accumulate in organisms through the food chain. When plastics come into contact with food, POPs can migrate into the food, impacting food quality and human health.
Due to the small size and unique physical properties, MNPs can cross biological barriers, entering the human body and potentially affecting multiple systems, including the digestive, immune, and nervous systems. MNPs can cause irritation and damage to mucous membranes after entering the gastrointestinal tract through the esophagus. Long-term ingestion of these microplastics can disrupt digestive system function and may even cause inflammation, ulcers, and other serious diseases. MNPs can carry pathogens such as bacteria and viruses into the human body [154]. Once these pathogens enter the body, they can trigger immune responses, leading to disease. The plastic particles themselves can also interfere with the normal functioning of the immune system, affecting its ability to resist diseases. Recent studies suggest that MNPs may have neurotoxic effects, damaging the nervous system [155–157]. These tiny particles can cross the blood-brain barrier and interact with protein fibers in neurons, affecting their normal function.
Source control is key to reducing plastic waste. By improving product design, production processes, and packaging methods, plastic usage can be fundamentally reduced. For instance, many countries around the globe have implemented bans on single-use plastics, resulting in significant reduction in plastic production [158]. Besides, several countries around the world have introduced plastic bottle deposit systems, to minimize the consumption of plastics. In China, since 2016, the growth of plastic products production and consumption decelerated, while the recycling rate has risen, surpassing 30% in 2021 [159]. Consumer education plays a crucial role in reducing plastic usage. By raising public awareness of plastic pollution and encouraging consumers to reduce plastic use and choose eco-friendly alternatives, a societal consensus and action can be formed. Finally, regulations and policies are important tools for promoting plastic pollution control. Strict regulations can limit the production and use of plastic products, promoting pollution control at the institutional level.
Effective plastic waste management requires addressing collection, recycling, and enforcement. Comprehensive management strategies and technical means are needed to achieve effective plastic waste management and resource utilization. The first step in plastic waste management is classification and collection. To enhance recycling rates, a well-established plastic waste classification and collection system is needed. Recycling and processing are key steps in resource utilization of plastic waste. Strengthening recycling facilities and improving processing efficiency are current important tasks. Enforcement and supervision ensure effective plastic waste management. Strengthening the supervision and enforcement of plastic waste treatment enterprises ensures compliance with regulations and prevents illegal dumping and pollution.
Plastic pollution in agricultural production is prominent. To promote green development and reduce plastic pollution, measures must be taken to regulate and manage the use of plastic products in agriculture. Strict regulations should be enacted to limit the use of plastic products in agriculture, especially non-degradable plastics such as mulch. Promoting eco-friendly agricultural alternatives effectively reduces plastic pollution. Efforts should be made to develop and promote the use of paper packaging, biodegradable materials, and other eco-friendly alternatives to reduce the use of plastics in agricultural production. Strengthening the supervision and enforcement of plastic product use in agriculture ensures the implementation of regulations. A robust regulatory system for plastic products like agricultural films should be established, with enhanced oversight from production to market quality.
The development of eco-friendly materials and technologies is vital for both environmental protection and economic transformation. Current development stages can focus on innovation, policy support, and public education to promote the development of sustainable materials and technologies. In contrast to conventional plastics, biodegradable plastics represent a potentially sustainable alternative for environmental protection and sustainability. These materials can decompose naturally in the environment under specific conditions, thereby contributing to the reduction of long-term waste accumulation [160, 161]. Natural fibers such as hemp, bamboo, sisal, and jute can serve as alternatives to plastics in various applications. Besides, these materials are renewable, biodegradable, and require low amount of energy to produce than petroleum-based plastics.
As MNPs pollution in the environment becomes increasingly prominent, research on their impact on the food production chain and potential health risks is critical. The widespread distribution and potential toxicity of MNPs have garnered global attention. To effectively address this challenge, comprehensive measures, as outlined below, must be taken at multiple levels to ensure food safety and quality.
Strengthening basic research is essential. MNPs have been detected in various human physiological systems, indicating their intrusion from the environment into the human body. Therefore, it is necessary to understand the behavior and mechanisms of MNPs in food production, processing, storage, and consumption, as well as their specific impacts on human health. This requires interdisciplinary collaboration involving researchers from environmental science, food safety, toxicology, and other fields.
Developing efficient detection technologies is necessary for ensuring food safety. Current methods for detecting MNPs in soil face limitations such as high technical requirements, limited resolution, and sample loss. Similar issues exist in MNPs detection in foods. Encouraging the development of efficient and accurate detection technologies for MNPs will meet the regulatory needs of food production and quality control.
Establishing stringent regulatory standards is key to ensuring food safety. As understanding of the health risks of MNPs deepens, clear limit requirements and corresponding regulatory measures must be established to prevent these pollutants from entering the food chain. This will help protect consumer health rights and promote the sustainable development of the food industry. Finally, strengthening international cooperation is necessary to address the challenge of MNPs pollution. This is a global issue requiring collective efforts from all countries. Through cross-border cooperation, sharing research findings, exchanging technologies and experiences, and jointly developing international standards, we can enhance the global food safety governance system. This will help humanity collectively address this challenge and ensure the safety and quality of food worldwide.
While food safety and public health are major concerns, toxins from plants have been identified as one of the key risks. These harmful substances can become part of the human diet in two ways, either through consumption of poisonous plants or indirectly via food contamination. Therefore, it is important to expand the knowledge on the different plant toxins, their effects, and how to mitigate them, so the safety and quality of the food supply can be guaranteed.
Plant toxins can be divided into several classes based on their chemical structure and their effect on human health. Some of the plant toxins are mycotoxins, alkaloids, oxalates, cyanogenic glycosides, glucosinolates and lectins.
Alkaloids are nitrogen-containing compounds that can have major effects on the nervous system. Some examples of common toxic alkaloids are glycoalkaloids such as solanine in potatoes [162] and tomatine in tomatoes [163]. High levels of solanine can cause symptoms ranging from nausea and vomiting to more severe neurological disturbances. Solanine nor tomatine has maximum limits (MLs) according to the legislation in force at EU. Tropane alkaloids (TAs) [164, 165] and pyrrolizidine alkaloids (PAs) have MLs according to EU legislation. TAs are naturally occurring compounds that serve as secondary metabolites in numerous plant species, particularly abundant in the Solanaceae family. More than 200 distinct TAs are synthesized by plants as a protective strategy against herbivores, pathogens, and competing vegetation. Methods to determine TAs (atropine, scopolamine, anisodamine, and homatropine) have been optimized and validated including QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) extraction followed by ultra-high performance liquid chromatography combined with time-of-flight mass spectrometry (UHPLC-ToF-MS) in the dried product of herbal infusions and in buckwheat and buckwheat products [166, 167]. PAs are a group of natural compounds found in several plant families, notably Boraginaceae, Asteraceae, and Fabaceae. These compounds are produced by plants as a defense mechanism against herbivores and are generally determined by LC-MS [168]. PAs pose significant health risks, primarily through liver damage, carcinogenicity, and genetic mutations. Chronic exposure can also lead to conditions such as pulmonary veno-occlusive disease (pulmonary hypertension), and reproductive toxicity [168].
Some glycosidic compounds, when hydrolyzed, produce substances associated with negative human health effects. This is the case of cyanogenic glycosides, found in cassava and almonds, that release cyanide when metabolized, posing a serious risk of poisoning if not properly processed. Hydrocyanic acid, including cyanogenic glycosides, has also defined MLs according to the EU legislation [169]. Cyanogenic glycosides such as lotaustralin, prunasin, taxiphyllin, and dhurrin have been determined using LC-MS/MS. Park et al. [170] identified the major dietary exposure sources and proposed the development of a preliminary risk assessment framework based on the dietary exposure assessment and the calculation of theoretical levels of hydrocyanic acid derived from cyanogenic glycoside concentrations. This is quite relevant in order to establish, in the near future, guidelines for the permissible intake of foods containing cyanogenic glycosides [170].
Other plant toxins are lectins, frequently found in beans and legumes. These compounds can interfere with nutrient absorption, namely protein, and cause gastrointestinal issues. Raw or undercooked kidney beans are particularly high in lectins, which can cause severe digestive disorders. However, these compounds are associated with plant defense, therefore lectin genes could be used for designing strategies for multigene transfer to generate resistance in susceptible crops [171]. Moreover, their utilization as potential antimicrobial agents for drug development and drug therapies has also been well reported [171].
Oxalates are other class of compounds found in plants that can have toxic effects in humans. They are present in foods like spinach and rhubarb. Physiologically, these compounds can bind to calcium, forming insoluble crystals that can lead to kidney stones and other health issues. Methods used to determine oxalate include electrochemical detection, LC-MS or GC-MS, enzymatic degradation of oxalate with oxalate oxidase and detection of hydrogen peroxide produced, and indicator displacement-based methods employing fluorescent or UV light-absorbing compounds [172].
Mycotoxins are fungal toxins that are found in crops. These compounds can contaminate plant-based foods like cereal grains and nuts [173, 174]. Aflatoxins, produced by Aspergillus species, are among the most potent carcinogens that can contaminate crops like peanuts and corn. EU legislation has established MLs for mycotoxins in diverse food products, however there are still many mycotoxins that do not have a maximum residue limit defined in food [169].
Despite their various health benefits, excessive or exclusive consumption of vegetables and seeds from the Brassicaceae family has been associated with several adverse health effects due to their high levels of glucosinolates. These include altered thyroid function and an increased risk of thyroid disorders, besides liver and kidney malfunction. These effects can also impair growth, reproductive performance, and may even be fatal. During chewing, the enzyme myrosinase converts glucosinolates into breakdown products such as thiocyanates and isothiocyanates, which have been associated to harmful impacts on human health [175].
Plant toxins can get into human food via various pathways, namely through 1) direct ingestion of toxic plants: for example, when cassava is improperly prepared it can cause cyanide poisoning; 2) post-harvest management: for instance, poor storage conditions such as improper drying and storage of crops can allow the growth of mycotoxins producing fungi; 3) processing and cooking: some cooking processes can decrease levels of plant toxins [175]; this is the case of boiling beans that reduces lectin content, making them safe for eating.
The health effects of plant toxins can vary widely depending on the type and amount of toxin consumed. Acute poisoning can result in immediate symptoms such as vomiting, nausea, diarrhea, and neurological symptoms. For example, chronic exposure to aflatoxins is a major risk factor for liver cancer, particularly in regions where staple foods like maize and peanuts are prone to contamination. Chronic exposure to lower levels of toxins can lead to long-term health issues, including liver and kidney injuries, cancer, and growth-related disorders. Long-term consumption of foods high in oxalates can contribute to the formation of kidney stones and other renal complications.
The guarantee of food safety involves a multi-faceted approach to managing plant toxins [176]. In this line, several strategies can be adopted to mitigate plant toxins. These include: 1) to develop plant varieties with lower toxin levels through traditional breeding or genetic engineering to reduce the risk of poisoning [177]; for example, cassava varieties with lower cyanogenic glycoside content have been developed; 2) to implement Good Agricultural Practices (GAP) to minimize contamination from non-edible crops to edible crops [178, 179]; 3) to carry out proper drying, storage, and handling of crops to prevent the growth of mycotoxin-producing fungi; this includes the use of appropriate storage facilities and monitoring moisture levels; 4) regular testing with suitable analytical methods and enforcement of regulations to ensure that contaminated products do not reach consumers; therefore, it is important that governments and regulatory bodies set up safety standards and monitor food products for plant toxins; 5) education of consumers about the risks associated with plant toxins and safe food preparation practices can significantly reduce the incidents with these compounds. Public health campaigns and educational programs are essential components of a comprehensive food safety strategy. This includes the education of consumers and food processors regarding safe preparation methods to reduce toxin levels in foods. For instance, soaking and boiling beans effectively reduces lectin content, making them safe for consumption.
Summing up, knowing the sources of plant toxins, together with their mitigation measures and effects, helps enhancing the quality and, consequently, the safety of foods. Further studying and innovating around plant breeding, agricultural practices, reliable analytical methods and processing will be very effective in addressing the risks of plant toxins. Moreover, creating consciousness among consumers regarding its safety can successfully reduce the effect of these natural compounds on public health.
Mass spectrometry (MS) and nuclear magnetic resonance (NMR) spectroscopy are among the main analytical techniques employed for targeted and untargeted studies in food science. Both techniques have advantages and limitations and in recent years are often used as complementary approaches to obtain the greatest amount of information on foods. This section discusses these two methodologies to determine organic contaminants in food, highlighting some distinctive aspects of their use for food authentication.
Organic contaminants in food have been mostly determined using liquid or gas chromatography coupled with MS (LC-MS or GC-MS) [180]. Representative legacy and emerging contaminants (ECs) are pesticides, veterinary drug residues, mycotoxins, polybrominated diphenyl ethers (PBDEs), other brominated flame retardants (BFRs), organophosphate esters (OPEs), short-chain chlorinated paraffins (SCCPs), per- and poly-fluoroalkyl substances (PFASs), polycyclic aromatic hydrocarbons (PAHs), microplastics, and so on [181]. Tandem quadrupole-MS employing multiple reaction monitoring (MRM) is a sensitive targeted method that plays an important role in detecting and quantifying organic contaminants commonly present at low concentrations [182]. High-resolution mass spectrometry (HRMS) has opened new perspectives in food contaminant analysis [183]. HRMS has changed the traditional analysis concept as it allows untargeted analysis (i.e., able to detect an unlimited number of known and unknown compounds, at least in theory) [184].
Food authentication is the process by which a food is verified as compliant with its label description [184]. It allows us to discriminate between food species, geographical origin, etc. Different analytical techniques are applied for food authentication, where in recent years ‘-omics’ approaches have been applied for a comprehensive understanding of food constituents [185]. MS is fundamentally applied in two fields proteomics and metabolomics [181], whereas NMR spectroscopy is mainly used in metabolomics [186–188].
Proton NMR-based metabolomics is used for food authentication:
16-O-methyl-cafestol and kahweol as specific markers of coffee Robusta [189, 190] and Arabica, respectively;
5-hydroxymethylfurfural, or 2-acetylfuran-3-glucopyranoside [191] as specific markers to authenticate honey samples, and
picrocrocin and glycosyl esters of crocetin are markers to authenticate saffron samples [192].
Similarly in olive oil adulteration with low-quality hazelnut oils, proton NMR analysis detected down to 10% adulterations [193].
Interestingly, proton NMR aided by chemometrics, was used to authenticate the Rochefort Trappist beers because of the two-fold higher pyruvic acid content in it compared to other types of beer [194].
The most effective analytical techniques for food authentication include NMR [186], MS, and hyphenated chromatographic methods (e.g., LC-MS, GC-MS, LC-DAD-NMR/MS). The hyphenated techniques enable high-throughput qualitative screening and discrimination of food characteristics. Discrimination among the samples is aided using both unsupervised pattern recognition algorithms [principal component analysis (PCA) and hierarchical cluster analysis (HCA)], and supervised pattern recognition algorithms [linear discriminant analysis (LDA), partial least squares discriminant analysis (PLS-DA), soft independent modelling of class analogy (SIMCA), and the Random Forests (RF) machine learning algorithm].
Proper usage and development of MS- and NMR-based spectral databases are essential to identify contaminants and their transformation products and to verify food authenticity and quality. Unfortunately, current databases lack comprehensive experimental data on all known contaminants, their transformation products, and overall food composition. In the case of MS, novel computational tools that predict MS fragmentation patterns in silico have been developed to assist with identifying contaminants for which tandem MS data are not available yet. The currently developed artificial intelligence (AI) and machine learning devices will be the future in this field [181].
The application of MS and NMR in food authentication provides a powerful and precise tool for verifying the origin and quality of food products, ensuring consumer trust and regulatory compliance. Widely shared standard protocols recognized by the international community need to be developed. Instruments operating at different parameters (magnetic field strengths in NMR, collision energies in MS), and sample preparation conditions make it difficult to compare results obtained in different laboratories. For this purpose, ring tests proposed by laboratories at the national level are emerging, which can pave the way for the creation of recognized protocols. Additionally, integrating AI and new imaging techniques presents further challenges related to data management, standardization, and development of robust algorithms [185, 195, 196].
Although the role of biosensors in the food sector has embraced different fields, e.g., pathogen sensing and GMO detection, the food authenticity field is still in an embryonic phase. Food authenticity can hold several dimensions, considering geographical origin, food composition and processing, that need to comply with what is provided to the consumer. Moreover, the main target of food adulteration is focused in high-value market products, such as wine, olive oil, animal products, among others. Most of these are based on the use of a specific cultivars/variety (plant species) or breed (animal species), which can be more easily identified using DNA-based markers, since these are not influenced by external conditions and remain stable throughout the food chain [197, 198].
Biosensors have gained attention in this field, especially due to their general interesting features: high sensitivity, reliability, portability, cost-effectiveness, and rapid response [199]. So, by using biomolecules, such as DNA, as a biorecognition element, numerous configurations can be designed targeting a given cultivar/variety/breed. Additionally, the fact that smaller targets may be used can help to overcome the limitations imposed by the high DNA degradation verified in highly-processed food products [200].
Even so, there is still a huge gap between the development of biosensing devices for other food applications and for food authenticity, mostly because of the difficulty of handling complex foods. When DNA extraction is required for a specific analysis, the use of efficient and quick DNA extraction protocols is imperative. Some research has been concentrated in designing devices that can select DNA, from such complex samples, through microfluidic approaches. Nevertheless, this requires sample preparation steps, that can increase detection time and costs. Thus, the search for direct sampling approaches is preferred.
Additionally, the biosensors’ analytical performance, considering quality and reliability of the obtained results, has been looked into. Through the use of new NMs, which are mainly used to modify the surface of the transducers, an increase in parameters, such as, sensitivity, selectivity, limit of detection (LOD), and limit of quantification (LOQ), can be improved, being considered nowadays a hot topic in this line of research [198, 201].
Most of the biosensors reported for authenticity purposes have been concentrated in animal species identification, either in meat samples or in derived processed products [198]. Some reports have been based on plant product e.g., authenticity along the wine chain (leaf, must and wine samples [197]), olive oil adulterant [202] and identification of species present in flours [203]. The determination of gluten in flours is relevant when dealing with celiac patients. Other food products are not so well studied, and would benefit from such a strategy, as the economic implication of fraudulent practices are substantial. The existence of more genomic information at affordable prices, gives way to a wider application of DNA-based methods for authenticity purposes. More, the progress observed in other technological fields, such as the development of new nanomaterials and technological solutions, can help to boost the development of biosensing devices that will be easier to use at a more affordable price, surpassing some of the still existent constrains.
The main objective of food analysis has always been food safety, but over time, factors pertaining to food quality, food traceability, food authenticity and processing have steadily grown in significance. The last decade researchers are moving from traditional procedures, which are characterized by targeting a small number of analytes and moderate analytical performance, to advanced methodologies that apply the most recent developments in food science as a result of rapid evolution of analytical instruments and techniques. Foodomics is a term that refers to a new discipline that studies the Food and Nutrition domains through the application and integration of advanced -omics technologies to improve consumer’s well-being, health, and knowledge. Foodomics technologies are highly useful for identifying the similarities and differences between food products and for determining the food fingerprint, which is a marker of food authenticity and quality [204–206]. This section offers a critical discussion of the most current advancements in foodomics technologies and chemometric approaches that are applied in this field.
In foodomics, the study of food and nutrition is integrated with -omics technologies and bioinformatic tools. The main -omics technologies used in foodomics include metabolomics, proteomics, and transcriptomics. A brief description of these technologies is provided below [207, 208].
The goal of metabolomics is to identify and quantify small molecules (typically < 1,000 Da) present in a biological system [209]. Metabolic fingerprinting and metabolic profiling are the two fundamental methods that can be applied in a global non-targeted metabolomics investigation. The goal of metabolic fingerprinting is to compare metabolite patterns that are altered in response to the biological environment, such as the reaction of the cell to a bioactive drug treatment. The approach of metabolic profiling is focused on examining a particular metabolic pathway or a family of related metabolites, such as carotenoids, flavonoids, and polyphenols [210]. However, it is impossible for a single methodology to assess the entire set of metabolites due to the chemical diversity and wide concentration dynamic range of metabolites (particularly in plants, where it is considerably bigger than in any other animal) [206]. High analytical sensitivity, shorter chromatographic run times, direct infusion techniques, scan speed and mass resolution are therefore the most important factors to consider when choosing a suitable methodology for a thorough metabolomic investigation [206, 211]. LC-HRMS, GC-HRMS or CE-HRMS, UHPLC-Q-Orbitrap-MS, UHPLC-Q-ToF-MS, direct infusion MS as well as NMR are the primary technologies utilized. In addition, ultra-HR-MS approaches based on flow-injection such as Fourier transform ion cyclotron mass spectroscopy (FT-ICR-MS) are also used to provide the food’s metabolome. Furthermore, the most widely used interfaces in HR-MS based metabolomics coupled to LC techniques are ESI (electrospray ionization) or heated electrospray ionization (HESI) sources, which operate in the positive ESI mode [206]. A broader metabolome coverage can be achieved by combining different analytical techniques, which yield complementing analytical information [212].
The extensive examination of a biological system’s proteome (set of proteins) at a certain moment is known as proteomics. It comprises protein identification and characterization [including post-translational modifications (PTM) and isoforms], their relative or absolute quantification, their position and how they interact with other molecules [213]. Recent developments in genome sequencing along with the advancement and refinement of instrumental analytical techniques have propelled proteomics forward. The creation of soft ionization techniques such as ESI and MALDI (matrix-assisted laser desorption/ionization) is one of this field’s most significant analytical developments [214, 215]. These techniques enable the ionization of proteins and/or peptides and the subsequent MS analysis of these substances. There are two ways that the proteomics can be performed, i.e., top-down and/or bottom-up approach. The top-down analysis is employed to characterize PTM found in proteins. It depends on the analysis of intact proteins that have been isolated by LC or two-dimensional gel electrophoresis and then detected by MS. Following mass analysis, the intact proteins’ ions are separated, fragmented in the collision cell, and subsequently identified by MS/MS [216]. The bottom-up approach, on the other hand, is distinguished by the enzymatic digestion of the proteins before their MS analysis [206, 217]. Following peptide/protein separation, the most widely utilized mass analyzers nowadays are Orbitrap® and quadrupole-time-of-flight (QTOF) [206]. Furthermore, the development of the latest Orbitrap Eclipse Tribrid MS with advanced quadrupole mass filter, dual-pressure linear ion trap and Orbitrap mass analyzers [218], as well as the advancement of ion mobility spectrometry (IMS)-based MS such as trapped IMS in timsTOF, T-wave IMS in SYNAPT G2-Si and field asymmetric IMS (FA-IMS) in Orbitrap MS are new trends in proteomics field [219–222].
Transcriptomics examines the transcriptome, which is a set of transcripts generated by an organism, tissue, or cell’s genes at a specific time. The primary goal of the transcriptome is to identify genes that are differentially expressed in response to a particular stimulus. The transcriptome is dynamic, with gene expression fluctuating in response to various stimuli [213]. Various high-throughput methods, such Massive Parallel Signature Sequencing and Serial Analysis of Gene Expression, have been utilized to examine the transcribed sequences [223, 224]. However, significant advancements in nanotechnology and microfluidics have made it possible to develop methods like massive RNA sequencing (RNA-Seq) and gene expression microarrays, which enable fast and effective analysis of thousands of transcribed sequences [225]. In recent years, there has been an increase in the use of gene expression microarrays for a variety of purposes, including the discovery of newly identified illnesses, the creation of novel diagnostic tools, and the identification of drug/food response systems as well as for foodomics applications [206].
Chemometric approaches for unsupervised and supervised pattern recognition, such as PCA, HCA, LDA, PLS-DA, SIMCA are widely used to handle complex foodomics data. The primary purpose of these tools is to evaluate classes, such as classifying samples as authentic or not authentic and adulterated or unadulterated. Apart from the qualitative chemometric tools, there are quantitative methodologies such as principal component regression (PCR), multiple linear regression (MLR) and partial least squares regression (PLSR) used as multivariate calibration tools [226]. In addition, the application of state-of-the-art machine learning tools such as support vector machine (SVM), artificial neural network (ANN) and RF has rapidly emerged in recent years [227].
Emerging food processing technologies, such as microwave heating, ohmic heating, moderate electric field, pulsed electric fields, cold plasma, high-pressure processing, and ultrasound, are poised to revolutionize future trends in Food Science and Foodomics. For example, ohmic heating has been proposed for sustainable green extraction that could provide sustainably high-quality extracts [228] and enriched with valuable bioactive compounds that could improve consumer’s well-being and health. Another example is the recently developed moderate electric field [229] to pasteurize milk (and potentially other liquid foods) at relatively low temperatures that extend the shelf-life with reduced environmental impact in terms of emitted carbon dioxide. Similarly, sonication improves bioactive compound extraction, extends perishable foods’ shelf life, and valorizes food waste [230]. These technologies can also assist bioprocesses such as fermentation [231]. Accordingly, industrial application of such novel food processing could produce novel ingredients and enhance the nutritional profile of foods. In foodomics, these technologies enable comprehensive profiling of food components, revealing insights into nutritional content and bioavailability. Food production based on such technologies could facilitate identifying and quantifying bioactive compounds, resulting in the development of functional foods designed for specific health benefits. These technologies collectively enhance food safety, quality, and sustainability, aligning with consumer demands for high-quality and safe foods. These could eventually facilitate the development of foods with targeted health benefits and advance the scientific information on the complex relationship between diet and health through foodomics.
The extraction of bioactive compounds from food by-products is an evolving field in line with the global push towards sustainability and circular economy. Traditional extraction methods often rely on chemical solvents, which pose significant environmental and health risks. In response, green extraction techniques have emerged that focus on reducing environmental impact and increasing efficiency. Green extraction techniques refer to a set of methods used to isolate components such as bioactive compounds from natural sources such as plants, food by-products and marine biomass, with a strong emphasis on environmental sustainability. These techniques aim to minimize the use of hazardous solvents, reduce energy consumption, and lower the overall environmental footprint of the extraction process. The principles of green extraction include the use of renewable resources, energy efficiency, waste reduction, and ensuring safety to environment and human health. Several green metrics tools are used to evaluate and ensure the sustainability of these extraction processes. These tools provide quantitative measures to evaluate the environmental impact, efficiency, and safety of extraction methods. Some of the key green metrics tools include E-Factor, Atom Economy, Energy Intensity, Carbon Footprint, and the most powerful and standardized tool, Life Cycle Assessment (LCA).
Some of the most widely used green extraction techniques in food analysis include microwave-assisted extraction, ultrasound-assisted extraction, enzyme-assisted extraction, compressed fluids’ extraction [supercritical CO2 and pressurized liquid extraction (scCO2 and PLE)], and the use of natural deep eutectic solvents (NADES). Among them, compressed fluids are particularly important because of their efficiency, selectivity, and low toxicity. Together, these techniques enhance the recovery and fractionation of bioactive compounds while minimizing the use of hazardous solvents and reducing environmental impact. In fact, these techniques can even be combined in biorefinery processes to maximize the type of bioactive compounds that can be extracted from a single biomass. By integrating these methods, the field of food science can achieve more sustainable and environmentally friendly practices, contributing significantly to the advancement of green chemistry and sustainable industrial processes, in line with the United Nations sustainable development goals (SDGs).
In order to illustrate interesting advances in this topic, some articles have been selected considering its contribution to green processes, sustainability and circular economy. For instance, Abderrezag et al. [232] explored the extraction of silymarin from Silybum marianum seeds by considering environmentally friendly solvents such as ethanol, CO2 and water to replace hazardous solvents such as hexane, and highlights their applicability in the food, pharmaceutical and cosmetic industries. The research highlights the benefits of gas expanded liquid (GXL) extraction, particularly using CO2-expanded ethanol. This technique significantly reduces viscosity and increases extraction efficiency, making it a viable industrial-scale method for obtaining highly polar compounds at lower pressures than those required for supercritical fluid extraction (SFE). This advancement helps reducing operating costs and environmental impact, furthering the shift toward sustainable practices in the extraction of bioactive compounds. The study “Extraction of neutral lipids and phospholipids from marine biomasses using subcritical and supercritical fluids” [233] is a key development in the utilization of marine biomass through green extraction techniques. This research investigates the use of scCO2 and subcritical dimethyl ether (DME) for the extraction of lipids and bioactive compounds from marine by-products. Both methods are highlighted for their ability to preserve valuable bioactive while minimizing energy consumption and environmental impact. A key finding is the superior performance of DME in extracting a full spectrum of phospholipids compared to scCO2 due to its partial miscibility with water and lower operating pressures. This study not only advances our understanding of the capabilities of green solvents, but also addresses regulatory and safety considerations to ensure that these methods are practical and compliant with industry standards.
One of the groundbreaking contributions to green extraction is the use of NADES as rising alternatives for food applications [234]. These are mixtures of two solids at room temperature that combine to form a liquid that can be used as a solvent. Composed of biocompatible and biodegradable components such as choline chloride and citric acid, NADES offer a non-toxic and environmentally friendly alternative to conventional solvents. The study, “Pressurized Natural Deep Eutectic Solvents: An Alternative Approach to Agro-soy By-products” [235] illustrates the potential of NADES in the extraction of phenolic compounds, flavonoids and isoflavonoids from soy by-products. Besides, in this work the solvent composition was optimized theoretically before extraction using solubility prediction software, in this case authors applied Hansen solubility parameters (HSPs), although other methodologies such as COSMO-RS (COnductor like Screening MOdel for Real Solvents) can be also employed. The study demonstrated the efficiency of NADES under pressurized conditions in combination with PLE. The PLE process at 120°C and 100 bar for 20 minutes yielded high concentrations of bioactive compounds, demonstrating a significant reduction in solvent consumption and energy use compared to traditional methods. Furthermore, Benvenutti et al. (2022) [236] used pressurized aqueous solutions functionalized with NADES, as a novel approach, to improve the recovery and protection of anthocyanins from Brazilian berry peel, a procedure useful for various food by-products. This integration of green solvents with advanced extraction technologies marks a significant step towards sustainable extraction processes.
Encapsulation methods have been emerging as appropriated alternatives to food industries, in order to increase the stability and the quality attributes to the processed foods. Encapsulated ingredients or additives consist of adding to a particular formulation the desirable components involved in a protective material (encapsulated agent), mostly biopolymers, that control the interactions of the inner component (core substance or biocomponent) with the environment.
The most common encapsulation materials used in food industries are natural polymers (like starch, carrageenan, chitosan, gelatin, sodium alginate, and others) or synthetic biopolymers [polyvinylpyrrolidone, poly(L-lactide), poly(lactic-co-glycolic acid), and others]. The selection of the encapsulation agent must be associated to its interactions with the core material and with the food system, and also to its resistance to the processing parameters, and its ability to protect the quality attributes for the necessary time. Besides, the encapsulated agent must be feasible with the process, allowing the adequate liberation of the core material to provide the necessary bioactivities and functional properties, without unnecessary increase in processing costs.
The encapsulation processes involve different systems like emulsions (macro, micro, nano), coacervate, gels and other complexes, combining the core material and the encapsulate agent, with the aid of solvents, co-solvents or anti-solvents, depending on the encapsulation mechanism applied.
There are several methods used for encapsulation, from classical procedures like spray-drying and extrusion, to coacervation, double emulsion, freeze-drying, fluidized bed coating, or alternative supercritical methods (anti-solvent, extraction of emulsion, solvent impregnation). The classical encapsulation methods can show some negative aspects such as poor control of the particle size and particle size distribution, low encapsulation efficiency, as well as some thermal degradation, depending on the solvents used. Supercritical encapsulation methods can provide a solution to some of drawbacks of the classical methods due to the increase in the number of variables (pressure, temperature, solvent/feed ratio, among others), providing a more “tunable” process, which allows better control of the particle’s formation. Also, these high-pressure methods can approach to a sustainable procedure due to the use of greener solvents (or anti-solvent), such as carbon dioxide, and lower temperature compared to some traditional methods. For instance, Andrade et al. (2020) [237] used the SEDS (Solution Enhanced Dispersion by Supercritical fluids) technique for the encapsulation of pink pepper extract, leading to spherical microparticles formation and encapsulation efficiency up to 95%. Besides, the SFE of emulsion (SFEE) combines the anti-solvent properties of supercritical carbon dioxide with the emulsion method, and was successfully used for the encapsulation of laurel leaves essential oil with 75% efficiency [238]. SFEE was also applied for the precipitation and encapsulation of the carotenoid fraction from pink shrimp residue in pluronic, and compared with the supercritical anti-solvent (SAS) method, where the SFEE provided 93% encapsulation, while SAS reached up to 74% efficiency [239]. Gas anti solvent (GAS) method with carbon dioxide was applied for the micronization of luteolin, providing a 10-fold size reduction, improving the antioxidant potential of the formed particles [240]. High-pressure methods were also applied to improve the stability of food grade colorants, such as the use of SAS method for the encapsulation of curcumin [241].
Therefore, the adequate selection of the materials and processes involved in the encapsulation of bioactive or functional components for food products can affect the properties and characteristics of the produced particles, such as morphology, size, particle distribution, and aspects related to functionality, stability, and bioavailability, and must be carefully considered for a specific application. Consequently, it is necessary to balance properly the costs involved, the safety of the product and also the benefits that an encapsulated agent can provide to a food product.
In conclusion, green extraction techniques and encapsulation strategies, supported by robust green metrics tools, represent a significant step towards sustainable industrial practices. These methods not only improve the efficiency of extracting valuable bioactive compounds from food by-products and other biomass, but also align with global sustainability goals by minimizing environmental impact and promoting the use of renewable resources. Encapsulation techniques further enhance the stability, bioavailability and controlled release of these bioactive compounds, ensuring their efficacy and extending their shelf life. Together, these advances provide a comprehensive approach to valorizing food by-products, contributing to a circular economy and promoting sustainable development in the food and pharmaceutical industries.
Food processing encompasses a wide range of techniques and methods applied to raw ingredients with the primary aim of improving food safety and convenience, shelf-life, and organoleptic properties. Food processing can generally be considered beneficial [242] by promoting the inactivation of food-borne pathogens or natural toxins, extending the shelf-life, or improving the digestibility and bioavailability of nutrients. It is expected that food processing has the minimum interference or improves the product’s palatability, taste, texture, and flavor of the product. It may also impart improved functional properties to the product. The impact of food processing on food microbiota is expected, since some of the technologies aim a microbiological control, and may affect not only the safety but also the microbial ecology of food.
In the era of the -omics tools, it is important to understand the impact of food processing on food bacterial communities, beyond the bacterial indicators of food safety. The number of studies on changes in food bacterial ecology due to food processing is still limited, with studies on the effects of food processing on the human gut microbiota being more common [243, 244]. The most classical food processing methods, such as thermal and chemical processing or fermentation, as well as the non-thermal methods (e.g., high-pressure, pulsed electric field, ultraviolet radiation, cold plasma, ozonation, ultrasound, membrane filtration), promote a modulation of the native bacterial communities [245–248]. This modulation of the bacterial communities due to the selective or stochastic removal of some of the native bacteria will affect the bacterial ecology of the product. The same can happen when external microbiota or antimicrobials are added to the food product, as is done with biopreservation methods [249]. These modulations of the native bacterial communities may or may not be beneficial. Removal of foodborne bacteria and increasing the abundance of probiotic bacteria are perhaps the most studied changes. However, the use of methods with selective effect may favor the survival of the most fitted or well adapted bacteria, including antibiotic resistant bacteria known to occur in food products [250–252]. Given the global health concerns about the rise of antibiotic resistant bacteria, the role of food processing in the spread and control of these bacteria deserves the full attention of the scientific community. Understanding and optimizing the effects of food processing on the microbiota of food products is essential to ensure the safety, nutritional quality, and health benefits of food.
The fruit and vegetable markets are constantly changing and seeking for new opportunities, in particular considering their capacity to provide nutrients and other chemicals required for human health. Unfortunately, the production of these foods is seasonal and usually perishable, making their processing necessary to increase shelf life [253]. The perceived extent and purpose of different processing techniques of edible foods follows the NOVA classification: unprocessed or minimally processed foods, processed culinary ingredients, processed foods, and ultra-processed groups. Independent from the type of process, there is always the possibility that the natural characteristics of the ingredients could be altered or lost through different mechanisms. One mechanism of special interests is the interaction between specialized metabolites, formerly known as secondary metabolites. Secondary metabolites are molecules derived from primary metabolites by biochemical transformation with macromolecules, including proteins, polysaccharides and lipids, which are building blocks of the structure and function of plant cells. The result of this chemical interaction may vary, but it likely leads to physicochemical, organoleptic, texture or functional changes of fruits and vegetables [254]. This section describes some of those interactions, both covalent and noncovalent, emphasizing on their actors and how the products change the nature of the processed fruit or vegetable.
Thermal or non-thermal processing has been associated with carotenoid degradation in vegetables. In fact, drying may result in 10–20% loss of carotenoids [255]. However, lipids may increase the bioavailability of carotenoids. As heating can break down cell walls, released carotenoids are able to mix with available or added lipids, improving absorption. Thus, this carotenoid-lipid interaction has been suggested as the most crucial event in bioavailability of carotenoids [256].
Cold plasma treatment is a novel non-thermal technology that helps maintaining the freshness and quality of fruits products without using chemicals [257]. The cold plasma, similar to ionized gas but at near room temperature, is generated by applying energy to a gas, creating a mixture of ions, electrons, and neutral particles. The reactive oxygen species induced during this approach have been linked to oxidation of phenolic compounds [258] leading to the formation of quinones, unstable intermediates that in turn can bind covalently to proteins to generate Lys/Cys/Arg-derived adducts [259], altering both the physicochemical and immunological characteristics of food proteins [260].
One of the most common chemical transformations that occurs during heating treatment of fruits is the Maillard reaction or non-enzymatic glycation, also known as the carbonyl ammonia reaction. It occurs by covalent interactions between carbonyl compounds, such as reducing sugars, unsaturated fatty acids, aldehydes and ketones, and amino chemicals, involving free amino groups of amino acids, peptides and proteins [261]. These chemical products serve for improvements in color, flavor, taste, as well as in antioxidant properties [262]. Unfortunately, industrial drying methods that include heating, such as hot-air and microwave-relate processes, in addition to produce high degrees of Maillard reaction, also generates advanced glycation end-products, proteins or lipids modified by covalent binding to sugars and their derivatives, such as glyceraldehyde, glycolaldehyde, methylglyoxal and acetaldehyde, that register several toxicity concerns [263].
Although there is a number of analytical techniques useful to study the formation of interactions between secondary metabolites and macromolecules, proteomics approaches have the capacity to provide insights in the nature of the derivatives formed, such as glycated proteins [264]. The understanding of these chemical processes will provide the basis for the production of functional fruit- and vegetable-derived products.
The World Health Organization recently published the bacterial priority pathogens list containing the most dangerous microorganisms for human health (https://www.who.int/news/item/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed). The list was divided into three main groups: 1) the “critical group” made up of pathogens with the highest warning to public health due to limited treatment options, high disease concern (mortality and morbidity), increasing trend in antibiotic bacterial resistance (ABR), and global mechanisms of resistance or strains in specific populations or geographical areas; 2) the “high-group” contains ABR pathogens significantly challenging to treat, with an increasing trend in resistance, high difficulty of prevention, high transmissibility and few potential treatments; 3) the so-called “medium group” ABR includes bacterial pathogens associated with moderate difficulty for treatment, and moderate trends in resistance, potentially representing a severe problem for some populations and specific geographical areas.
Given the increased antibiotic resistance developed over the years by microorganisms, the scientific world is in a frantic search for new substances, even those of natural origin, which, through their biochemical composition in terms of primary and specialized metabolites (proteins/peptides, enzymes, polyphenols, flavonoids, carotenoids, anthocyanins, volatile component), can somehow make up for the weakness of conventional antibiotics, and capable of helping science also by limiting, or even blocking, bacterial virulence. Moreover, naturally sourced and formulated products are perceived to be generally safe compared to synthetic drugs, with fewer and short-term side effects [265]. As a result, there is growing awareness and acceptance of all-natural products. The mechanisms through which these substances, molecules or cocktails of natural molecules, act against pathogens can be multiple. They, for example, can act by modifying some properties of bacteria, such as its hemolytic activity (which is an essential parameter for some bacteria, such as Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa); they can also modify the hydrophobicity of the bacterial cells, thus affecting its capability to adhere to the human cells (i.e., the gut cells, or the lung cells), or block the quorum-sensing mechanism of bacterial cell-cell communication [266]. Some studies reported an important contribution of such compounds in inhibiting, or at least in reducing, the capacity of pathogens in forming the so-called biofilm, complex communities of microorganisms, including bacteria, growing on surfaces and embedded in a self-produced matrix of extracellular polymeric substances; or act when this is mature and lead to genotypic and phenotypic modification of the bacterial cell/community which determine an increase in the bacterial virulence. Generally, the study of bacterial biofilm is done through now standardized analytical techniques, which can evaluate whether natural substances are able to limit bacterial adhesion processes, or mature biofilms, and whether they have the capacity to influence microbial metabolism, limiting those processes of transformation that lead to an increase in virulence. The crystal violet assay is commonly employed to assess the effects of extracts or molecules on biofilm formation. Additionally, the MTT colorimetric method [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] can be particularly useful for studying how potential anti-biofilm agents influence the metabolism of planktonic cells trapped within the biofilm environment. The development of new anti-biofilm agents requires a comprehensive systems-level understanding of these mechanisms, as well as the discovery of new mechanisms. First step of study assumes in vitro biofilm model that should provide a method of assessing the relative activity of a group of compounds and relating these to other compounds in the literature and assess the probability that specific compounds will work against biofilms in a relevant circumstance. The study can be supported by -omics approaches such as transcriptomics, metabolomics, proteomics, and in silico techniques, which can also be integrated to better understand biofilm biology [265, 267]. The need for increasingly rapid results has led to the use of new techniques for the microbiological sector. Through the so-called biofilm electrostatic test (BET) it is possible to reproduce the ability of bacteria to form biofilms through electrostatic interactions with a pyroelectrified carrier. The system can produce various types of biofilms in a much shorter incubation time (2 hours). Therefore, due to its speed, simplicity, and cost-effectiveness, it is well suited for rapid and easy implementation in a microbiology laboratory [268].
Natural compounds, as well as synthetic drugs, should be tested in biologically relevant biofilm models to increase the likelihood that these candidate agents selected from in silico approaches are effective in humans.
Numerous reviews discussed the harmful effects of biofilm formation in the food industry, highlighting how biofilms contribute to the presence of spoilage microorganisms and foodborne pathogens. These contaminations are often associated with the presence of biofilms in processing facilities. Waste materials from the agri-food industry can play an important role in combating microbial biofilms and offer valuable benefits in preventing or reducing infections and biofilm formation, even on food surfaces. For instance, fruit peels, like those from Citrus fruits, which are high in polyphenols and precious oils, have shown strong antibiofilm properties [269, 270].
The study of bacterial biofilms has been supported by the possibility of using cultured and differentiated human organoids (categorized into three primary forms: air-liquid interface (ALI) models, 3D spheroid organoids, and organoid-on-a-chip models [271]) capable of recapitulating, to a large extent, the histological and physiological characteristics of the native organs, for instance, the native human mucociliary epithelium, to monitor P. aeruginosa biofilm [272] or human epidermis organoid model to study the S. aureus biofilm [273]. The use of organoids is still rarely applied to studying natural compounds [274], but their versatility will be certainly seen a huge interest for new future studies on the antibiofilm properties of natural compounds.
Food packaging is one of the main issues to consider to stabilize a food product, since packaging is a requirement to maintain quality and safety of products subjected to food processing technologies. In most of these technologies, the package is designed to provide a passive barrier that limits the negative interaction of the external environment on food, including gases (oxygen, carbon dioxide), vapours (humidity, aromas or odours), micro- and macro-organisms, radiations, etc. Furthermore, packages should be inert to the food, and should not release substances onto nor adsorb components from the food that may affect its quality and/or safety.
Food packaging science and technology has evolved much as a response of the needs of both industry and consumer demands. At present, a novel emerging technology is being researched and slowly implemented in the industry of packaging converters and in the food industry. Smart packaging technologies involving active and intelligent packaging technologies are at the forefront of the present innovations. Another issue of high relevance is the environmental impact of packaging, especially important in flexible packaging with plastic materials, which is being addressed by the preparation of biodegradable materials or by the introduction of recycled materials.
Active packaging is considered one of the so-called food emerging technologies, since in many cases, it is one of the hurdles to reduce or limit the incident of spoilage processes. Active packaging systems include those designs in which the packaging components interact with the food or its surrounding headspace, causing a beneficial effect such as reducing oxidation rate [275], reducing ripening rate [276], or microbial growth [277]. Most of the successful commercial systems are based on independent devices that contain substances that retain water, oxygen, ethylene, and improving product stability. Similar devices have been also designed to release substances in the headspace such as SO2, flavouring components, antioxidant or antimicrobial molecules. Nevertheless, the possibility of breakage of these devices by manufacture failure or by consumer manipulation may result in product contamination and potential intoxication, and there is a push towards the development of packages that incorporate the active substances in the polymeric walls of the package. The formulation of active materials has few but relevant requirements: the active agent may change the plastic properties, release or sorption depends on plastic permeability, the active capacity requires of an effective triggering mechanism, and the final design should consider the specific requirements of the food product (concentration of the active substance, kinetics and thermodynamics of the action [278]). Also, the potential sensorial effect of the release active substance on the product must be considered, since often these molecules have pungent aroma. Many reports, especially in antimicrobial and antioxidant packaging have been published but most of them are difficult to implement, or show activity in vitro but not under real conditions. Efficient active packaging must provide a valuable enhancement in quality, safety, and/or shelf life. In this regard, the improvements in stability achieved through the use of active packaging also result in more sustainable packaging, despite the increase in materials required. This has been demonstrated by life cycle analysis studies, which show that the reduction of food waste is the greatest contribution of packaging to reducing environmental impact [279]. More effort is needed to fully develop this technology which can be very helpful to reduce food losses.
In intelligent packaging there are two distinct lines: informative and shelf-life indicative. The former is very relevant for traceability, stock controls, corporative image, overture assurance, and it is being implemented very rapidly in the food industry. They are mainly based on barcodes, QR codes, radiofrequency identification (RFID) labelling, near filed and augmented reality technologies. Shelf-life indicators are not so common. They are mainly conformed by a pigmented dot that suffers a colour change as response to a condition that takes place in the food or package headspace, such as humidity, pH, oxygen presence or diverse metabolites such as sulphur or nitrogen containing molecules [280]. Although many reports have been written, especially those based on anthocyanins that are pH sensitive, many are not-implementable, or describe conditions that rarely occurs in real food systems. Future trends in this line should include, easily visible colour changes that are correlated to the food spoilage process.
Although packaging is a fundamental part of food technology and in ensuring the quality and safety of food, the final packages are seen as something worthless to throw away and that accumulate in landfills and in nature due to improper disposal. Thus, many efforts are focused on the three R: Reducing material consumption, Reusing packages and Recycling materials. During the last 3 decades, the amount of material to make any type of packaging has been significantly reduced by wall thinning and improved 3D design, in the so-called Eco-Design (as little as possible as much as necessary principle) [281]. Metal and glass packages have been reused and recycled for decades. Recycling of metal and glass requires the melting of the materials in furnaces at a very high temperature so most organic contamination and the new material is as good as the one of first use. Another question is how expensive is the process in terms of money, energy or environmental impact. In this regard, the use of LCA tools can provide relevant information [282].
Plastic recycling or reuse is not that simple due to several issues. First, conventional petrochemical polymers (polyethylene, polypropylene, polyesters or polyamides) are amorphous or semicrystalline materials that allow mass transfer processes of low molecular weight molecules, including permanent gases (O2, CO2), vapors (H2O, food aroma or external odor components), colored molecules and residues and additives present in the plastic matrix. These mass exchanges might be controlled in their first use as packaging material but are unknown in reused or recycled materials. Most polymers are converted at low to medium temperatures (from 80 to 250°C) so that organic permeants, sorbates or migrants are not fully removed from the polymer matrices. Second, all plastics are very similar in aspect but not in chemistry what makes very difficult their selection as postconsumer residues for appropriate recycling, and their mixture commonly reduces the value of the final recycled material. Third, with a few exceptions, some bottles, trays and films, most flexible packaging is made of multilayer structures composed of various polymers, whose separation is impractical and their recycling invalid. Thus, in practice these materials are not recycled, or they are used as a composite of a non-food application, or they are incinerated with energy recovery thanks to their high combustion energy value. Thus, instead of circular economy, plastic materials should approach an In-Spiral economy as efficient as possible, where recovered materials can be incorporated together with other inputs [283].
Diverse world-wide regulations are being addressed for the environmental impact reduction of plastic packaging. Two principal lines are being developed: the use of compostable materials as alternative to traditional non-sustainable/non-degradable plastic materials or the reduction of package complexity to favor recycling processes. The use of compostable packaging materials resolves the issue of recycling, since after consumption, these packages are disposed with organic residues and used in composting. In this regard, several compostable and bio-based materials have been developed for processing by conventional conversion technologies (extrusion, coextrusion, injection, etc.) such as thermoplastic starch, polylactic acid, polyhydroxyalkanoates, cellophane, by themselves or combined with petrol-based compostable polymers such as polycaprolactone, polybutylene succinate, polybutylene succinate adipate, or polybutylene adipate terephthalate [284]. However, packaging and food companies are resilient to implement these materials because they are more expensive and still, they have to pay taxes for their postconsumer processing. Besides this, some LCA comparisons are questioning their reduced environmental impact [285]. Recycling appears to be the preferred process for plastic packaging. Indeed, EU regulations are promoting the inclusion of a minimum percentage of recycled materials in food packages. Many improvements have made recycling suitable for second use as packaging materials, such as better cleaning and deinking processes, monomaterial structures, cleaner mechanical and chemical recycling processes [286]. Nevertheless, the presence in the packaging materials of non-intentionally added substances will increase with the incorporation of recycled polymers and often migration tests will be required to assess food safety. Lastly, and by no means less important, it is essential to remember that packaging is key to reducing food losses, facilitating transportation, and that without it, modern society would not exist. The reduction, recycling, and compostability of packaging materials must not compromise the primary functions of packaging, as its use is the main means of preventing food loss, which, as previously mentioned, is responsible for the greatest environmental impact.
Currently, more than ever, consumers, particularly elderly people, are aware of health benefits of phytochemicals in plant-based diets including polyphenols, vitamins, proteins, unsaturated fatty acids, carotenoids, phytosterols, among others, due to their bioactive properties associated to preventive effects against certain diseases such as cancer, cardiovascular and neurodegenerative diseases. The demand is for foods with potential health benefits or with health claims, rather than only tasty or aromatic foods, or processed foods subjected to industrial and/or domestic processing. Nevertheless, although a great number of bioactive compounds can be found in foods and several other natural sources, only a small proportion will be absorbed, metabolized, and used to exert their biological function. However, to exert their beneficial effects, it is essential that the bioactive compounds are accessible to be used and/or stored in the body. For this reason, it is important to take into consideration both the bioaccessibility and bioavailability of bioactive constituents [287].
Despite being different, the concepts of bioaccessibility and bioavailability, are often cause of confusion. The bioaccessibility represents the fraction of a compound released from the food matrix during digestion and becoming available for absorption into the intestinal lumen and reaching the blood stream to be delivered to the target tissue. This is the mandatory condition for bioavailability and bioactivity (Figure 3).
Schematic presentation of the different steps included in bioavailability and bioaccessibility
Several parameters and physiological conditions can influence the extent of bioactive compounds bioaccessibility including the composition of food matrix in terms of nutrients, particle size, food processing (thermal vs non-thermal processing), the digestive enzymes which determine the extent of liberation of bioactive compounds from food matrix, and the pH conditions through the digestive system. On the other hand, the bioavailability as a measure of the extent of absorption and utilization of bioactive compounds by the human body [288], in its active form and in appropriate concentration, is influenced by the chemical nature of the compounds in terms of lipophilicity and hydrophilicity, the stereochemistry and molecular structure (size and complexity) of the compound which can affect the capacity to pass through the intestinal mucosal barrier, by the presence of other substances/nutrients that can enhance or inhibit the extension of absorption, in addition to individual unmodifiable physiological factors such as gender, age, genetics, gut microbiome and health status [289]. In this context, the bioavailability is crucial to their potential beneficial effects on human health.
Several in vivo (usually carried out in animals or humans) and in vitro methodologies have been developed for analysing the bioaccessibility and bioavailability of bioactive compounds. Although the in vivo studies provide relevant data related to bioavailability and high scientific evidence, they present several limitations in terms of experimental design, and ethical constraints, they are normally long-term studies, in addition to difficulties in data interpretation. On the other hand, the in vitro studies are usually employed as cost-effective, fast and reproducible step in analysing the bioaccessibility of bioactive compounds, however it does not mimic the in vivo processes. Moreover, ex vivo, and in situ methodologies are also different models that allow to study bioaccessibility and bioavailability of bioactive compounds.
There exists a wide array of methods to evaluate the bioaccessibility and bioactivity of natural bioactive components. In general, the method of choice is selected depending on the target bioactivity being studied. A vast number of in vitro methods to study different bioactivities have extensively been applied in the field. Although bioactive compounds may show promising results, it is of utmost importance to confirm if the studied bioactivity can take place in vivo. In fact, the digestion process may significantly influence the chemical nature of the bioactive components, thus, being able to positively or negatively affect the bioactivity observed in the native structure [290]. Moreover, the bioactive compounds should exert their bioactivity in the final target tissue. For these reasons, the study of their bioaccessibility and bioavailability is necessary. In vitro gastrointestinal models include static protocols, which main example is the INFOGEST method [291], and dynamic studies. Static protocols are generally simpler although they are designed to simulate the complex conditions within the human digestion process. As static protocols are widespread, comparisons among samples are even possible. On the other hand, dynamic models are closer to reality, allowing longer times, recovery of samples and the online fermentation using human microbiota. Depending on the model, oral, stomach, small intestine and colon compartments can be used, each of one containing their own enzymes, chemicals and process conditions to mimic real gastrointestinal digestion [292–294]. In addition, interestingly, new models including absorption studies, for instance using Caco-2 cells, are being implemented [295].
However, the most interesting and ideal data can be obtained from human volunteers [296]. Nevertheless, these in vivo studies are costly, difficult to be implemented and present ethical constraints. Moreover, the obtained data could even be hampered by the great inter-individual variability that exists. For these reasons, other in vivo models based on animals are also applied. Even considering all the related difficulties, in vivo studies should be used, when possible, at least, for the validation of the in vitro models [297].
The absorption mechanisms which take place in the intestinal lumen are one of the most important factors affecting the bioavailability of ingested food bioactive compounds. These complex mechanisms are influenced by several factors including the physicochemical properties of bioactive compounds, related with its structural complexity and polymerization degree, stereochemistry, stability and solubility, and the interactions with the gastrointestinal environment. The low-molecular-weight polyphenols (depending on the class) are, normally, absorbed in the small intestine, while the oligomeric and polymeric polyphenols remain unabsorbed in the small intestine and reach the colon almost unchanged where are subjected to extensive catabolism into a series of low-molecular-weight phenolics in the colon through enzyme-catalysed reactions by the gut microbiota transforming them into absorbable metabolites [298].
The most important mechanisms involved in the absorption of bioactive compounds include: 1) passive diffusion, in which the lipophilic compounds are absorbed across the intestinal epithelium; 2) facilitated diffusion; and 3) active transport. The first two mechanisms involve diffusion towards a concentration gradient through the intestinal cells into the blood circulation. The latter mechanism works against the concentration gradient and can result either in the increase of compounds in the blood circulation or in the efflux of the ingredients back to the intestinal lumen [287, 299].
Since the bioactive compounds do not have the optimal physicochemical properties necessary for passive diffusion, their permeability is improved by using trans‐membrane transporters, through uptake and efflux mechanisms. Several transporters are involved in the uptake and enhance of bioactive compounds across the intestine including glucose transporters family, human sodium glucose transporters family and organic anion transporters [287, 300].
Different encapsulation tools may be used to effectively protect the bioactive compounds during food processing, storage and also during the first stages of gastro-intestinal digestion, helping these components to reach the small intestine or the gut, where they may be liberated, intact. It has been observed that particles with an average size greater than 100 µm are not easily absorbed in the small intestine, while the greatest absorption rate occurs in nanometric particles [301]. For this reason, nanoencapsulation tools are more interesting. Nanoencapsulation allows obtaining particle sizes between 10 and 1,000 nm [302]. Among the available published tools, NEs [303], liposomes [304], solid lipid NPs [305], nanostructured lipid carriers [306], ethosomes [307], phytosomes [308], niosomes [309], glycerosomes [310] or invasomes [311] are highlighted. The use of these techniques must be coupled to encapsulating agents that are the compounds responsible for the protective effect on the bioactive compounds. Hydrocolloids, different carbohydrates, cellulose, proteins and lipids have been employed in nanoencapsulation [312]. In the case of lipidic or low polarity bioactive compounds, liposome-based encapsulation processes have become widespread [313]. Thanks to their amphiphilic behaviour, phospholipids, forming a monolayered closed structure, are chosen as carrier compounds, as they are considered safe to be employed in the food industry [314]. Nano-liposomal encapsulation has emerged as an exceptionally effective approach, with capsules containing bioactive compounds reduced to nanoscale for enhanced efficacy.
Understanding food-gut microbiota interactions will become increasingly important in the years to come, as gut bacteria can metabolize nutrients and bioactive compounds into potential health-promoting molecules [315–317]. Revealing food-gut microbiota interactions has the potential to revolutionize our understanding of human health and to create a more personalized and effective approach to nutrition [318]. Food-gut microbiota interactions have a profound impact, opening new avenues in various fields: 1) personalized nutrition and 2) food technology. Firstly, deciphering how individual gut microbiota metabolize different foods allows for personalized dietary recommendations. This can lead to more effective disease prevention and management tailored to individual needs, designing prebiotics to promote beneficial bacteria, designing foods to modulate specific gut functions, or even developing new therapeutic approaches for gut-related diseases. Secondly, by understanding how different food components influence gut health, we can create new food products that promote gut health, improve nutrient absorption, and even prevent specific diseases. To tackle this challenge, both in vitro and in vivo models are valuable tools for understanding food-gut microbiota interactions [319, 320]. In vivo models are considered crucial because they involve living organisms and, therefore, provide a more holistic and physiologically relevant context for studying these interactions. However, they have limitations, such as ethical considerations, housing conditions, and differences in gut microbiota composition across species. In vitro models are recognized as cost-effective and efficient ways to explore hypotheses about these interactions. Still, they are limited by their need for epithelial and immune compartments and challenges in culturing strict anaerobic microorganisms.
As soon as the complex interactions between what we eat and the trillions of bacteria that live in our gut and intestinal cells are revealed in a controlled environment, the biological role of gut microbial metabolites (GMMs) can be further investigated. During the last decade, intestinal organoids (IOs) have become increasingly relevant in the food field. IOs are 3D cell cultures grown from human tissues that mimic the structure and function of the intestinal epithelial cells [321, 322]. The main advantage of IOs compared to traditional methods, such as animal models and immortalized cell cultures, is that traditional methods do not capture the full complexity of these interactions. Thus, IOs offer a more accurate and controlled environment for studying these interactions, paving the way for a better understanding of the role of GMMs in human health and disease. Over the past decade, researchers have developed methods to create both basal-out and apical-out organoids [323], each with its advantages and limitations. Basal-out organoids, while useful, pose challenges due to their internal lumen, requiring careful consideration of polarity in experiments [315, 324–327]. Apical-out organoids allow better access to the lumen-facing side but come with issues related to scalability and mixed polarity [323]. Recently, 2D IO monolayers have been developed to mimic intestinal physiology more closely. These monolayers have shown promise in studying drug metabolism and absorption but still require further optimization and development for broader applications [328]. While organoid technology has greatly advanced our understanding of gut biology, ongoing research is necessary to optimize these systems for greater applicability in studying foodomics applications. In addition, multi-omics approaches, such as metabolomics and proteomics, should be used in conjunction with IOs to paint a complete picture of the complex interplay between diet, the gut microbiome, and the host at the cellular level.
Nutraceuticals can be defined as biologically active components obtained from food or other natural sources that, when delivered in a non-food matrix, provide health benefits beyond basic nutrition. Nonetheless, these products lack an official and generally accepted definition, and are usually marketed as dietary supplements, co-existing in a vague zone between pharmaceuticals and food [329]. Many nutraceuticals lack sufficient scientific evidence regarding their efficacy and safety. Consequently, recent trends and challenges in this field focus on understanding their interactions with biological systems and the mechanisms behind their health benefits.
One major trend is the development of advanced delivery systems, particularly nanotechnology-based approaches, to improve the bioavailability and efficacy of nutraceuticals [330, 331]. Another significant area of research involves the use of computational tools and chemoinformatics to design and develop next-generation nutraceuticals. These methods enable researchers to better understand the chemical interactions and biological pathways involved, leading to more effective and targeted nutraceutical formulations [330].
The assessment of the effects and mechanisms of action of bioactive compounds is difficult to address in humans owing to their cost, complexity and ethical constraints. Animal models can be used for a comprehensive evaluation of their effects, however, experiments in mice and other mammals are expensive and time consuming and are also subject to ethical limitations. In this respect, the nematode Caenorhabditis elegans (C. elegans) has emerged as an amenable platform to be used as an animal in vivo model. This non-pathogenic worm possesses a series of convenient features, such as small size, short life cycle, transparent body, easy handling, and low maintenance and propagation costs. As an invertebrate, there are no ethical constraints on its experimental usage. Its fully sequenced genome (9.7 × 107 bps, ~19,000 genes) shows more than 60% homology with humans. Information about gene functions is freely available through WormBase (https://wormbase.org/#012-34-5), and many relevant metabolic pathways are conserved between C. elegans and humans, including those involved in processes such as aging, metabolism, apoptosis, cell signalling, and cell cycle. There is also a wide array of loss-of-function mutants and transgenic strains containing reporter gene fusions available for purchase, and genetic manipulation is possible through biotechnological technologies such as RNA interference knockdown, CRISPR-Cas9 editing, and auxin-inducible degron technology, allowing forward and reverse genetic screening.
Recently, this organism has been used for the discovery and efficient evaluation of bioactive compounds, elucidation of mechanisms of action, and identification of biological targets. It also serves as a platform for initial activity screening to obtain preliminary information, leading to more documented and cost-effective late preclinical developments in mammal models. A significant number of articles have been published over the last two decades using C. elegans to assess food components in modulating physiopathological processes such as aging, oxidative stress, inflammation, fat metabolism and deposition, and neurodegeneration [332, 333].
The role of the gut microbiota in the bioavailability and effects of food components is another hot topic in current research. Metabolism by the gut microbiota appears to be a key factor in explaining the large inter-individual variability in the biological response to bioactives. It is known that the composition and functionality of the microbiota vary among individuals, which has led to the definition of particular ‘metabotypes’ regarding gut metabolism capacity and its further influence on the effects of food components in the organism, as demonstrated for compounds such as isoflavones, lignans, and ellagitannins. The main factors driving the prevalence of metabotypes among populations are unclear, although age, sex, background diet, and disease state are likely to have an influence [334]. Different methodological approaches have been used to study the interplay between diet and gut microbiota. Gastrointestinal simulation can help identify microbial metabolites and define the microbial communities involved in the metabolism of food components. Animal models may be useful for relating bioactivity at the systemic level with the composition or functionality of the microbiota. Human nutritional intervention trials help examine the determinants and distribution of metabotypes and allow for a more holistic approach to assessing the impact of bioactives on disease-related biomarkers [335].
Overall, the integration of nanotechnology, computational methods, and gut microbiome research is paving the way for innovative nutraceutical products with enhanced health benefits and more precise therapeutic applications. However, challenges remain, particularly in ensuring the safety and efficacy of these advanced delivery systems and understanding the complex interactions within biological systems [329–331, 335].
Health can be represented as an optimal status point in a multidimensional space whose dimensions may coincide with the individual oxidative, inflammatory, and metabolic profiles, together with other pathophysiological parameters [335]. Many cofactors contribute to orienting movements in this space and, among the nutritional ones, the intake of an effective dose of the bioactive components present in the diet is crucial. In this context, the food matrix effect acquires a primary role, since the structure of a food, even with the same chemical composition, can influence the intake and bioavailability of nutrients and other bioactive components, leading to large differences in the biological impact. In fact, epidemiological studies have been undertaken recently, which highlight the harmful effects of food processing when pushed to extreme levels, such as completely bringing unstructured food [336]. Consequently, food consumption requires an objective coding system for classification, including food structure as a further descriptor of quality. So far, the classification has mostly grouped raw foods, i.e., not cooked or processed, into more than thirty different categories based on compositional data. It is clear now, how far this classification is from the actual descriptive need for the diet, and nutritional tables, in addition to listing the various nutrients, should provide structural and rheological information. This need is increasingly relevant, since the food industry is trying to generate new, more sustainable foods, with the intention of replacing traditional, higher impact food with something from alternative sources, and claiming to imitate sensorial properties and bioavailability of nutrients [337]. An emblematic case is that of vegetable burgers prepared by extrusion of vegetable or fungal proteins. For this reason, any technology capable of capturing the structure of the matrix is fundamental for any future food classification. Lately, there have been attempts to classify foods by considering the effects of processing on the original matrix structure, e.g., the NOVA system mentioned in section Interaction of the secondary metabolites with macromolecules in fruit and vegetable processing. However, the attempt has stopped at the level of the number of processing steps and the list of additives included in the formulation, often used to ensure a texture acceptable to the consumer [338]. The limitation of the NOVA system pays the price of the great difficulty in using comprehensible and classifiable descriptors to characterize the matrix structure. With these purposes, NMR spectroscopy can be of great help, when used within the foodomics approach, being able to go well beyond other analytical techniques, limited to the description of the mixture as a compositional table. In fact, having the power to observe matter in its functional entirety, the NMR analysis can be used to extract supramolecular information, define anisotropy parameters of heterogeneous mixtures, and study the flows of substances between the compartments that characterize the complex systems found in many foods [339]. MALDI MS imaging also has great potential in food analysis to provide relevant spatial information on the distributions of a wide range of compound classes in different matrices, including fresh and processed foods. To this end, machine learning tools for MS data analysis to study the penetration level of analytes into depth from irregular surfaces are of great benefit [340].
Usually, foods are not simple homogeneous mixtures since the structure of the food matrix is profoundly influenced by the close coupling between ingredients of different natures present in the various formulations and the technologies applied. Food structure, food texture, nutrient digestibility and consumer product preferences and choices are intrinsically linked. Food products are made largely of carbohydrates, proteins and lipids, forming clusters that behave like pseudo-molecules of higher molecular weight than the individual constituent molecules, resulting in heterogeneous materials composed of structural elements or domains, such as bubbles, drops, threads, and particles [341]. Complex food structures are formed due to the multiple interactions that proteins, lipids and polysaccharides establish under different conditions in an aqueous, such as in starch, gelatines, or oily medium. In addition to water, other structural elements can be identified in foods at a supramolecular structure level, such as oil droplets, gas cells, fat crystals, strands, granules, micelles and interfaces. The structural elements, in various combinations and proportions, can exist in different states (glassy/rubbery/crystalline/liquid and solubilized) even at uniform temperatures and water activities.
The dimensions/size and shape/form of the structural elements, i.e., particles, strands and pores, create the different textural properties of the food products [342]. This structural heterogeneity will necessarily influence the molecular dynamics of the system and consequently the macroscopic attributes of food quality and their behaviour during storage. Physically separating reagents into microstructural locations can control biochemical activity by preventing substrates and enzymes from coming into contact.
Stability can be fully grasped only if food molecular dynamics and structure are taken into consideration, i.e., an appropriate understanding of the behaviour of food products requires knowledge of its composition, structure and molecular dynamics, through the 3D arrangement of the various structural elements and their interactions [343].
The technological operations underlying the production of food are often accused of destroying the structure of the natural matrix that constitutes the raw materials, a fact considered deleterious for the quality of the food. The matrix integrity, indeed, is seen as an intrinsically positive element, which deteriorates during processing of ingredients into edible foods. It has been suggested that the presence of cell-free nutrients, as well as the lack of fermentable fibre and phytochemicals, could be responsible for intestinal dysbiosis [344].
The destruction of the barriers that compartmentalize animal tissues and, even more, plant tissues, makes nutrients immediately available for absorption, creating blood sugar spikes that can overload normal physiological functions, thus generating systemic inflammation of low grade [345]. For this reason, in consideration of its disintegrating capacity, fractionation is the most incriminated practice in food processing. As for sugars, also for lipids the gastrointestinal fate depends on their structural organization in the matrix [346]. When oil droplets are dispersed in a solid food matrix (e.g., cheese or yogurt), the structure of the surrounding food matrix becomes the dominant factor controlling digestion, influencing the bioavailability of oil-soluble vitamins. Increasing oil droplet size reduces bioaccessibility by inhibiting lipid digestion and reducing micelle solubilization. In protein-rich foods such as cheese, disintegration of the protein network occurs primarily at the gastric and intestinal levels, thus subordinating the subsequent release of fat aggregates to matrix degradation [347]. Intact cell wall structure and protein matrices are impervious to amylase and can prevent or slow the diffusion of the enzyme into the substrate. The starch-protein interaction in white flours could explain a decrease in the glycaemic response in vivo as well as a reduction in digestibility in vitro, such that the removal of gluten from wheat flour induces a high glycaemic index in bread without gluten.
Meat analogues are a class of food products that imitate the sensory attributes of meat products but are produced with proteins from more sustainable sources, e.g., plant protein isolates, that are subjected to extrusion or shear-cell technology. In these products, the presence of other food ingredients or components, such as lipids and polyphenols, may affect protein digestibility. These effects are still poorly understood for the lack of knowledge of the matrices.
Several techniques can be applied to measure the structure of food materials either directly (optical and confocal microscopy, tomography, scanning and electron microscopy) or indirectly from measurements of the mechanical response or by NMR spectroscopy.
One way to define the structure of the food matrix is to map the distribution of water within the compartments determined by the different structural elements that originate from the other molecules that contribute to the construction of the matrix. The confinement of water in delimited spaces, in contact with compartmental walls with different hydrophilic balance, reflects on water molecular dynamics and its chemical exchanges, giving it chemical-physical characteristics that make such molecule different, depending on the chemical environment, when observed using NMR. In other words, not only does the inhomogeneous distribution of water content between different volume units (voxels) provide information on the structure of the food matrix, but also the state (free/bound) and dynamics of water are important to provide relevant information on the structure. Measuring changes in the distribution and mobility of water represents a significant step in predicting the stability of a food, since the “availability” of water within the matrix profoundly influences the chemical, physical and microbiological quality of foods. The dynamics of water mobility can be an indirect measure of the limits to the free diffusion of water molecules exerted by the matrix compared to a barrier-free solution. The restrictive constraints on free diffusion can explain the consequent inhibition of the participation of water in reactions in which it participates as a reagent, for example, hydrolysis, or as a solvent, allowing the diffusion of substrates [348].
To measure water distribution and its associated properties by NMR, three possible paths are possible: 1) the nuclear relaxation time is the generally measured parameter, being associated with the rotational and translational correlation times, mediated by chemical exchange, of the molecules that have some degree of freedom in their movements within a rigid matrix; 2) immediately contiguous to this parameter, the magnetic resonance images, normally consisting of a density map of protons filtered through their nuclear relaxation time, are exploited. However, little information emerges regarding the “chemical distribution of molecules” within the delimited spaces (voxels). 3) The future challenge will consist in the use of spectroscopic techniques, containing information on the chemical shift, to draw maps over the scanned space, within which the sample is placed [349]. This information is certainly more difficult to measure without exploiting the rotation at the magic angle, which however has the task of eliminating the effects of anisotropy. It will be the task of the new data analysis tools, based on machine and deep learning, to extract coherent information from very noisy data, where part of what is normally considered noise can become information if appropriately processed.
Chronic diseases are the major public health problems in Western countries, and in addition thoroughly undermining the sustainability of health care systems. These diseases are responsible for 92% of all deaths in Europe, with cardiovascular diseases (41%) and cancer (28%) being the major causes [350]. Improved care, combined with a growing aging population, places significant managerial and economic pressures on National Health Systems. In particular, the increasing prevalence of patients with multiple conditions (one-third of the adult population and more than two-thirds of the elderly population) poses multiple challenges, including the establishment of Treatment Guidelines for these patients, as well as the identification of outcome parameters that consider clinical complexity. A focused approach to complexity will allow optimization of therapeutic proposals, thus moving into a scenario of responsive and personalized medicine capable of improving the cost-effectiveness of interventions [351].
Natural bioactive compounds can be: 1) essential for human health due to their multiple biological effects, such as reduction of non-communicable diseases (NCDs) risk factors, including cardiovascular and metabolic diseases, antioxidant, antimutagenic, anticarcinogenic, anti-allergic, anti-inflammatory and antimicrobial activities; 2) useful as food additives to maintain food quality, safety and stability and to improve shell life, palatability and attractiveness; 3) useful as food supplements or nutraceuticals to correct nutritional deficiencies, maintain adequate nutrient and macro and micronutrient intake, support physiological functions or prevent risk factors affecting health [352].
A better understanding of the links between biodiversity, health and disease represents an important opportunity for policy development and can improve our understanding of how they are interconnected: health-focused measures influence biodiversity and conservation measures influence health.
Diverse ecosystems are rich in a variety of plant species, which serve as sources for potential pharmaceutical compounds [353]. For instance, the periwinkle plant (Catharanthus roseus), native to Madagascar, has provided key alkaloids used in cancer treatments like vincristine and vinblastine [354]. Recent studies discuss [355, 356] the importance of biodiversity in the discovery of medicinal compounds. The decline in biodiversity can limit access to these valuable resources, particularly for indigenous and local communities that rely on traditional medicine for health care. When plant species disappear due to habitat destruction, climate change, or overharvesting, communities may lose vital medicinal knowledge and practices [357]. Biodiversity is crucial for food plants, impacting food security, nutrition, and resilience in agricultural systems. By supporting a diverse range of food plants, we can enhance nutrition, improve agricultural practices, and ensure food security in the face of environmental challenges. Protecting and promoting biodiversity in food crops is vital for a sustainable future [358]. Medicinal and food plants often attract a variety of pollinators, such as bees and butterflies. These pollinators are essential for the reproduction of many plants, including those that are crucial for food security. By supporting pollinator populations, medicinal plants help maintain the health of entire ecosystems [359].
The breadth and complexity of these relationships emphasize the need for an integrative, multidisciplinary and systemic approach to the health of people, livestock and wildlife in the context of the ecosystem. Loss of biodiversity, habitat fragmentation and the loss of natural environments threaten the full range of life-supporting services provided by ecosystems at all levels of biodiversity, including species, genetic and ecosystem diversity [360]. The disruption of ecosystem services has direct and indirect implications for public health, which are likely to exacerbate existing health inequities, whether through exposure to environmental hazards or through the loss of livelihoods. The concept of ‘One Health’ now represents a strategic model for the development of mutually beneficial policies and interventions between human health and biodiversity [361].
The One Health holistic vision, or health model based on the integration of different disciplines, is both ancient and current. It is based on the recognition that human health, animal health and ecosystem health are interlinked inseparably. It is officially recognized by the European Commission and all international organizations as a relevant strategy in all areas that benefit from collaboration between different disciplines (physicians, botanists, nutritionists, veterinarians, environmentalists, economists, sociologists, etc.) [362].
All of this, however, is struggling to happen, the One Health concept has been significantly undermined, often due to economic interests that prioritize short-term profits over the long-term interconnected health of humans, animals, and ecosystems, as recently highlighted by Mumford et al. [361]. This has led to policies and practices that have neglected the interconnectedness of human, animal and environmental health. For instance, industrial farming can promote zoonotic diseases, while environmental degradation can exacerbate health issues [363]. Addressing these challenges requires a holistic approach that considers the broader implications of our choices, fostering collaboration among sectors to promote sustainable practices that prioritize overall well-being. The One Health approach can significantly benefit from “high throughput” technologies found in -omic sciences, such as genomics, proteomics, metabolomics, and transcriptomics. These technologies enable large-scale data collection and analysis, providing insights into complex biological systems and play a crucial role within the One Health framework by providing comprehensive data that links human, animal, and environmental health, improving the ability to understand and address health problems holistically, opening the way for more effective prevention and intervention strategies. Researchers could better predict and mitigate health risks, ultimately leading to more effective disease prevention and control strategies. This holistic approach fosters collaboration across disciplines, encouraging a comprehensive understanding of health that considers all interconnected factors. As an example, the evolution of measurement and complex data integration systems, explored and adapted to research activities in the field of oncology (where it is now possible to discern in the enormous genetic and epigenetic heterogeneity of neoplastic cells some patterns of information that are significant and therefore useful for the purposes of personalized therapies), now allows their adoption in other areas, such as chronic NCDs. The goal is to integrate experimental evidence (genome, transcriptome, proteome, metabolome, exposome, etc.) with clinical-epidemiological evidence to generate predictive models that can, down to the level of the individual patient, serve as stratifies for optimizing prevention, diagnosis and treatment [364].
Biochemical (and physiological) analysis of cell behavior, read through a quantitative analysis of the metabolic asset, allows the acquisition of information relevant to understanding the deregulatory events that insist on the cell in pathological states.
NMR and MS based -omics technologies, (metabolomics, proteomics and lipidomics) have enabled the molecular level systems biology investigation of organisms in unprecedented detail [365, 366]. These methods have proven useful in identifying biomarkers that, when combined with more strictly clinical ones, could significantly increase the levels of accuracy and specificity of the same, both in the diagnostic field and in the follow-up of specific treatments. These -omics technologies allow the evaluation of complex biological systems, as well as the mechanisms of bioactive food compounds or the nutritional status and the exposures to environmental stressors that affect them [367, 368] (Figure 4).
Interactions between multiomics and the exposome in humans and animals. EWAS: exposome-wide association study; GWAS: genome-wide association study; MWAS: microbiome-wide association study (reproduced with permission from Gao [368], Environmental Science & Technology, 2021, 55, 2790–2799; Figure 4; © 2021 American Chemical Society)
Foodomics is an emerging field that focuses on the comprehensive study of food and its components, particularly how they interact with the gut microbiome and influence health [369]. Foodomics analyzes the nutritional and bioactive components of food, including vitamins, minerals, phytochemicals, and other compounds that may influence gut microbiota that plays a critical role in metabolizing food components, influencing nutrient absorption, and producing metabolites that can affect health [204]. Foodomics helps identifying how specific foods or dietary patterns shape microbial communities, promoting the growth of beneficial bacteria, which in turn produce metabolites like short-chain fatty acids (SCFAs) that have anti-inflammatory and health-promoting effects. Understanding these interactions is key to developing dietary recommendations and health outcomes (such as obesity, diabetes, and gastrointestinal diseases). The insights gained from foodomics can lead to personalized dietary recommendations and nutritional protocols based on individual microbiome profiles, promoting better health outcomes tailored to specific needs [370, 371]. The One Health approach and foodomics are complementary fields that enhance our understanding of health by recognizing the interconnectedness of human, animal, and environmental health, particularly in the context of food systems. By integrating foodomics within the One Health framework, we can better understand the complex relationships between diet, health, and the environment, leading to more effective public health strategies and sustainable food systems.
The concept of One Health merges biodiversity, food and human health. Biodiversity on Earth ensures the quality and quantity of food, after which, the food impact human health greatly [372]. This holistic approach underscores the profound impact of biodiversity loss on food systems and consequently on human health, presenting both challenges and opportunities for future sustainability.
Biodiversity is crucial for maintaining resilient ecosystems and sustainable food production systems [373]. Agricultural biodiversity is fundamental for ensuring food security in the face of changing environmental conditions and evolving dietary needs. Diverse ecosystems provide essential services such as pollination, pest control, and nutrient cycling. Biodiversity loss, driven by human activities such as habitat destruction, climate change, and pollution, poses a critical threat to global food security [374]. The diversity of crops, livestock, marine and freshwater supports agricultural resilience and productivity, while monoculture systems and overexploitation of natural resources diminish genetic diversity and ecosystem health, increasing vulnerability to crop failures, pests, and diseases. The biodiversity loss reduces dietary diversity, exacerbating malnutrition and diet-related health issues, particularly in communities reliant on diverse local foods. Addressing these challenges needs comprehensive strategies that integrate biodiversity conservation with sustainable agricultural practices and policies, ensuring resilient food systems capable of meeting future global food demands while preserving ecological integrity and human well-being [375, 376].
Healthy ecosystems provide a myriad of services that are essential for human health and well-being. These ecosystem services include clean air and water, regulation of climate and disease, and cultural and aesthetic benefits. Biodiversity loss disrupts these services, increasing the risk of waterborne diseases, air pollution-related illnesses, and exposure to infectious diseases. Furthermore, biodiversity loss can impact the availability and nutritional quality of food.
The link between biodiversity loss and human health is increasingly recognized in scientific research. Loss of biodiversity can contribute to the rise and spread of zoonotic diseases, which are infections transmitted between animals and humans. The destruction of natural habitats and increased human-wildlife interactions create opportunities for pathogens to spill over from wildlife reservoirs to human populations. Moreover, biodiversity loss can have indirect effects on human health through changes in food availability and nutritional quality. Reduced agricultural biodiversity may limit dietary diversity, leading to deficiencies in essential nutrients and contributing to diet-related diseases such as obesity and malnutrition.
In Latin America, the One Health approach is particularly relevant due to the region’s rich biodiversity, diverse food systems, and significant health challenges. This continent is home to a wide variety of native crops and animal species, which play a crucial role in local diets. Promoting the use of these biodiverse food sources can enhance nutrition and food security. Another very relevant aspect is related to traditional knowledge: indigenous and local communities often possess traditional knowledge about sustainable food practices and medicinal plants. Integrating this knowledge into health and nutrition strategies can improve health outcomes and preserve cultural heritage. Many Latin American countries are experiencing a nutrition transition, with increasing rates of obesity and NCDs [377]. One example is the importance that ancient crops still play in feeding populations and local development. These traditional crops contribute to agricultural biodiversity, which is essential for the resilience and sustainability of ecosystems. They are an integral part of the cultural identity and culinary traditions of many Latin American communities. Reviving and promoting these crops can help addressing modern nutritional challenges, such as obesity and malnutrition, by providing diverse and nutrient-rich food options [378, 379]. A One Health approach can address these issues by promoting local, nutrient-dense foods and sustainable agricultural practices. By prioritizing sustainable land and water management, Latin America can create a more resilient food system that supports both biodiversity and the health of its populations. This holistic approach is essential for tackling the intertwined challenges of food security, environmental degradation, and public health. This concept should also increasingly be kept in mind in view of the tropical deforestation and agricultural development in Latin America are closely intertwined issues that pose significant challenges for the environment, biodiversity, and local communities. One of the primary drivers of tropical deforestation is the expansion of agriculture, particularly for commodities like soy, palm oil, coffee, sugarcane and cotton. The demand for these commodities often comes from global markets, creating a complex challenge for sustainability. Addressing the impacts of these industries requires coordinated efforts involving government regulation, sustainable agricultural practices, and consumer awareness to promote responsible sourcing and conservation. These activities often lead to the clearing of vast areas of rainforest. The study by Pendrill et al. [380] found that, between 2011 and 2015, the tropics lost from 6.4 to 8.8 million hectares of forest per year to agricultural land [380]. Foodomics has the potential to significantly enhance the understanding of food systems in Latin America, promoting better health outcomes and sustainable practices. By integrating traditional knowledge with modern scientific techniques, foodomics can help preserve biodiversity, improve nutrition, and create economic opportunities, ultimately leading to healthier communities and a more resilient food system.
2D: 2-dimensional
3D: 3-dimensional
ABR: antibiotic bacterial resistance
AI: artificial intelligence
ANM: analytical nanometrology
ANN: artificial neural network
ANT: analytical nanotechnology
BET: biofilm electrostatic test
BFRs: brominated flame retardants
CEC: capillary electrochromatography
DME: dimethyl ether
ECs: emerging contaminants
ESI: electrospray ionization
EU: European Union
FA-IMS: field asymmetric ion mobility spectrometry
FFF: field flow fractionation
FITC: fluorescein isothiocyanate
FT-ICR-MS: Fourier transform ion cyclotron mass spectroscopy
GAP: good agricultural practices
GAS: gas anti solvent
GICA: gold immunochromatography assay
GMMs: gut microbial metabolites
GXL: gas expanded liquids
HCA: hierarchical cluster analysis
HDC: hydrodynamic chromatography
HESI: heated electrospray ionization
HRMS: high-resolution mass spectrometry
HSPs: Hansen solubility parameters
I.D.: internal dimension
IMS: ion mobility spectrometry
IOs: intestinal organoids
LCA: life cycle assessment
LDA: linear discriminant analysis
LOD: limit of detection
LOQ: limit of quantification
MALDI: matrix-assisted laser desorption/ionization
MLR: multiple linear regression
MLs: maximum limits
MNPs: microplastics and nanoplastics
MS: mass spectrometry
MW: microwave
NADES: natural deep eutectic solvents
nano-LC: nano-liquid chromatography
NCDs: non-communicable diseases
NEs: nanoemulsions
NLCs: nanostructured lipidic carriers
NLs: nanoliposomes
NMR: nuclear magnetic resonance
NMs: nanomaterials
NOMs: nanostructured organic materials
NP: nanoparticle
nSPR: nano-amplified surface plasmon resonance
OPEs: organophosphate esters
OT: open tubular
PAHs: polycyclic aromatic hydrocarbons
PAs: pyrrolizidine alkaloids
PBDEs: polybrominated diphenyl ethers
PCA: principal component analysis
PCR: principal component regression
PEF: pulsed electric field
PFASs: per- and poly-fluoroalkyl substances
PLE: pressurized liquid extraction
PLS-DA: partial least squares discriminant analysis
PLSR: partial least squares regression
POPs: persistent organic pollutants
PTM: post-translational modifications
QCM: quartz crystal microbalance
QTOF: quadrupole-time-of-flight
RF: Random Forests
RFID: radiofrequency identification
RNA-Seq: RNA sequencing
SAS: supercritical anti-solvent
scCO2: supercritical CO2
SCCPs: short-chain chlorinated paraffins
SCFAs: short-chain fatty acids
SDGs: United Nations sustainable development goals
SFE: supercritical fluid extraction
SFEE: supercritical fluid extraction of emulsion
SIMCA: soft independent modelling of class analogy
SLNs: solid lipidic NPs
sp-ICP-MS: single-particle ICP-MS
SVM: support vector machine
TAs: tropane alkaloids
UHPLC-ToF-MS: ultra-high performance liquid chromatography combined with time-of-flight mass spectrometry
US: ultrasound
EI and AC: Supervision (coordinated this work), Writing—original draft and Writing—review & editing (elaborated a first scheme, wrote the Introduction and revised the different contributions). CB, MF, and DKR: Writing—review & editing (revised the first and the final drafts of this work and proof-reading). F Capozzi: Writing—original draft (wrote section Foodomics and NMR spectroscopy to better define food matrix impact on the bioaccessibility of nutrients and bioactive compounds). YC: Writing—original draft (wrote section Analytical nanotechnology and nanoscience in food science). F Coppola and FN: Writing—original draft (wrote section Antibiofilm capacity of natural compounds). SF: Writing—original draft (wrote section The use of nano-liquid chromatography in food analysis). SRSF and JAM: Writing—original draft (wrote section Green extraction techniques and encapsulation strategies of bioactive compounds from food by-products). MG: Writing—original draft (wrote section Novel processing technologies). RG: Writing—original draft (wrote section Smart packaging and the potential effects of the introduction of recycled plastics in food packaging). MH, JR, and JSC: Writing—original draft (wrote section Bioactive compounds and their bioaccessibility and bioavailability). XL and CK: Writing—original draft (wrote section Microplastics and nanoplastics: analysis of their impact on food production and quality). LM and YP: Writing—original draft (wrote section Determination of organic contaminants in food safety and authenticity based on target and non-target MS and NMR methodologies). PML: Writing—original draft (wrote section Biosensors for food authenticity assessment). DDN and CP: Writing—original draft (wrote section New trends in foodomics for food authentication). JOV: Writing—original draft (wrote section Interaction of the secondary metabolites with macromolecules in fruit and vegetable processing). LR: Writing—original draft (wrote section A perspective from Europe about One-Health: biodiversity, nutrition and human health and A perspective from Latin America about One-Health: biodiversity, nutrition and human health). ÁR: Writing—original draft (wrote section Analytical nanometrology in food science). ASS: Writing—original draft (wrote section Contamination of foods with plant toxins). CSB and JSR: Writing—original draft (wrote section New trends and challenges in the interaction of nutraceuticals with biological systems and last advances in our understanding of the mechanisms behind their health benefits). IVM: Writing—original draft (wrote section Effect of food processing on the food microbiota). ZZ: Writing—original draft (wrote section A perspective from China about One-Health: biodiversity, nutrition and human health). All authors read and approved the submitted version.
Elena Ibáñez and Alejandro Cifuentes are the Editors-in-Chief of Exploration of Foods and Foodomics (EFF). The other authors are Editorial Board Members of EFF. All the authors were not involved in the decision-making or the review process of this manuscript.
Not applicable.
Not applicable.
Not applicable.
Not applicable.
José S. Câmara was supported by FCT-Fundação para a Ciência e a Tecnologia through the CQM Base Fund, [UIDB/00674/2020], Programmatic Fund, [UIDP/00674/2020], and by ARDITI-Agência Regional para o Desenvolvimento da Investigação Tecnologia e Inovação, through the project M1420-01-0145-FEDER-000005—Centro de Química da Madeira—CQM+ (Madeira 14-20 Program). Xianhua Liu was supported by the grant from the NSFC [#42377380] on data collection and analysis. Ángel Rios thanks for the support given by Spanish Ministry of Science and Innovation grant number [PID2022-138761NB-I00], and by Junta de Comunidades de Castilla-La Mancha grant number [JCCM SBPLY/21/180501/000188]. Paula Martins-Lopes thanks to FCT- BioISI center grants (DOI: 10.54499/UIDB/04046/2020). Francesco Capozzi thanks the support from the National Recovery and Resilience Plan, Mission 4 Component 2 Investment 1.3 - Call for tender No. 341 of March 15, 2022, of Italian Ministry of University and Research funded by the European Union—NextGenerationEU; Award Number: Project code [PE00000003], Concession Decree No. 1550 of October 11, 2022, adopted by the Italian Ministry of University and Research, [CUP D93C22000890001], Project title “ON Foods - Research and innovation network on food and nutrition Sustainability, Safety and Security – Working ON Foods”. Ana Sanches Silva was supported by [UIDB/00211/2020] with funding from FCT/MCTES through national funds. Luca Rastrelli was supported by the following project: “National Biodiversity Future Center - NBFC” funded under the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.4 - Call for tender No. 3138 of December 16, 2021, rectified by Decree n.3175 of December 18, 2021, of Italian Ministry of University and Research funded by the European Union – NextGenerationEU, Project code [CN_00000033], Concession Decree No. 1034 of June 17, 2022, adopted by the Italian Ministry of University and Research, [CUP D43C22001260001]. Ivone Vaz-Moreira was also supported by funding from the National Funds from FCT - Fundação para a Ciência e a Tecnologia through project [UIDB/50016/2020]. Yolanda Picó was supported by Grant [RTI2018-097158-B-C31] funded by MCIN/AEI/10.13039/501100011033 and “ERDF A way of making Europe” and by the Grant [CIPROM/2021/032] by the Generalitat Valenciana. Filomena Nazzaro thanks the support from: 1) the National Recovery and Resilience Plan, Mission 4 Component 2 Investment 3.1 “STRENGTHENING THE MIRRI ITALIAN RESEARCH INFRASTRUCTURE FOR SUSTAINABLE BIOSCIENCE AND BIOECONOMY” – (SUS-MIRRI.IT) supported by the Italian Ministry of University, Project code [IR0000005 – SUS-MIRRI – D.D n. 3264 del 28/12/2021 - DD MUR n. 114 del 21/06/2022 - CUP CNR D13C22001390001], and 2): the National Recovery and Resilience Plan Project “National Research Centre for Agricultural Technologies, Agritech”, Spoke 8 “New models of circular economy in agriculture through the valorization and recycle of wastes” [CUP B83C22002840001], supported by the Italian Ministry of University. Dilip K. Rai was supported by the Irish Department of Agriculture, Food and Marine under the grant [DAFM2021R616] (Flavoromics of Grass-Fed Beef and Lamb). Alejandro Cifuentes, Elena Ibañez, Jose Antonio Mendiola and Miguel Herrero were supported by grant [PID2020-113050RB-I00] funded by MCIN/AEI/10.13039/501100011033 (Spain). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
© The Author(s) 2024.
Copyright: © The Author(s) 2024. This is an Open Access article licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, sharing, adaptation, distribution and reproduction in any medium or format, for any purpose, even commercially, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
