-
79 results in Exploration of NeuroscienceSort byLatest
 Celebrating the legacy of Russel J. Reiter: a pioneer in melatonin researchOpen AccessEditorialDirk M. Hermann, Ertugrul KilicPublished: March 24, 2025 Explor Neurosci. 2025;4:100679
Celebrating the legacy of Russel J. Reiter: a pioneer in melatonin researchOpen AccessEditorialDirk M. Hermann, Ertugrul KilicPublished: March 24, 2025 Explor Neurosci. 2025;4:100679
DOI: https://doi.org/10.37349/en.2025.100679
This article belongs to the special issue Identification of Therapeutic Targets in the Pathogenesis of Neurological Diseases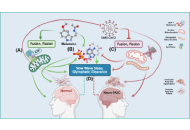 Melatonin regulation of phase separation in Neuro-PASC: out-maneuvering Janus-faced amyloidsOpen AccessReviewThe SAR-CoV-2 virus has evolved to co-exist with human hosts, albeit at a substantial energetic cost resulting in post-infection neurological manifestations [Neuro-post-acute sequelae of SARS-CoV-2 [...] Read more.Doris Loh, Russel J. ReiterPublished: March 24, 2025 Explor Neurosci. 2025;4:100678
Melatonin regulation of phase separation in Neuro-PASC: out-maneuvering Janus-faced amyloidsOpen AccessReviewThe SAR-CoV-2 virus has evolved to co-exist with human hosts, albeit at a substantial energetic cost resulting in post-infection neurological manifestations [Neuro-post-acute sequelae of SARS-CoV-2 [...] Read more.Doris Loh, Russel J. ReiterPublished: March 24, 2025 Explor Neurosci. 2025;4:100678
DOI: https://doi.org/10.37349/en.2025.100678
This article belongs to the special issue Identification of Therapeutic Targets in the Pathogenesis of Neurological Diseases Raw resting-state electroencephalogram biomarker emerges as an indicator of Alzheimer’s disease over a two-year periodOpen AccessOriginal ArticleAim: Alzheimer’s disease (AD) is associated with several electrophysiological biomarkers. These biomarkers are associated with global decline in cognition and a diagnosis of AD. However, a spec [...] Read more.Ezra C. HolstonPublished: March 24, 2025 Explor Neurosci. 2025;4:100677
Raw resting-state electroencephalogram biomarker emerges as an indicator of Alzheimer’s disease over a two-year periodOpen AccessOriginal ArticleAim: Alzheimer’s disease (AD) is associated with several electrophysiological biomarkers. These biomarkers are associated with global decline in cognition and a diagnosis of AD. However, a spec [...] Read more.Ezra C. HolstonPublished: March 24, 2025 Explor Neurosci. 2025;4:100677
DOI: https://doi.org/10.37349/en.2025.100677
This article belongs to the special issue Alzheimer's Disease Mechanisms of astrocytic and microglial purinergic signaling in homeostatic regulation and implications for neurological diseaseOpen AccessReviewPurinergic signaling, mediated by ATP and adenosine receptors, plays a crucial role in cellular communication and homeostasis within the central nervous system (CNS), particularly by regulating syna [...] Read more.Moawiah M NaffaaPublished: March 11, 2025 Explor Neurosci. 2025;4:100676
Mechanisms of astrocytic and microglial purinergic signaling in homeostatic regulation and implications for neurological diseaseOpen AccessReviewPurinergic signaling, mediated by ATP and adenosine receptors, plays a crucial role in cellular communication and homeostasis within the central nervous system (CNS), particularly by regulating syna [...] Read more.Moawiah M NaffaaPublished: March 11, 2025 Explor Neurosci. 2025;4:100676
DOI: https://doi.org/10.37349/en.2025.100676
This article belongs to the special issue Purinergic Signaling in Neuroinflammation and Pain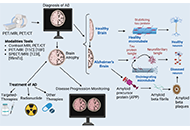 The role of neuroimaging in Alzheimer’s disease: implications for the diagnosis, monitoring disease progression, and treatmentOpen AccessReviewAlzheimer’s disease (AD) is a neurodegenerative disorder that affects millions of people worldwide. It presents a significant challenge in terms of accurate diagnosis, disease progression monitori [...] Read more.Julius Mulumba ... Yong YangPublished: February 25, 2025 Explor Neurosci. 2025;4:100675
The role of neuroimaging in Alzheimer’s disease: implications for the diagnosis, monitoring disease progression, and treatmentOpen AccessReviewAlzheimer’s disease (AD) is a neurodegenerative disorder that affects millions of people worldwide. It presents a significant challenge in terms of accurate diagnosis, disease progression monitori [...] Read more.Julius Mulumba ... Yong YangPublished: February 25, 2025 Explor Neurosci. 2025;4:100675
DOI: https://doi.org/10.37349/en.2025.100675
This article belongs to the special issue Alzheimer's Disease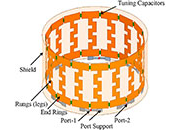 Neurostimulation devices to treat Alzheimer’s diseaseOpen AccessReviewThe use of neurostimulation devices for the treatment of Alzheimer’s disease (AD) is a growing field. In this review, we examine the mechanism of action and therapeutic indications of these neuros [...] Read more.Felipe P. Perez ... Maher RizkallaPublished: February 25, 2025 Explor Neurosci. 2025;4:100674
Neurostimulation devices to treat Alzheimer’s diseaseOpen AccessReviewThe use of neurostimulation devices for the treatment of Alzheimer’s disease (AD) is a growing field. In this review, we examine the mechanism of action and therapeutic indications of these neuros [...] Read more.Felipe P. Perez ... Maher RizkallaPublished: February 25, 2025 Explor Neurosci. 2025;4:100674
DOI: https://doi.org/10.37349/en.2025.100674
This article belongs to the special issue Alzheimer's Disease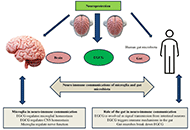 Neuroprotective insights into epigallocatechin gallate (EGCG) for neurodegenerative disordersOpen AccessReviewNeurodegenerative disorders, including Alzheimer’s, Parkinson’s, Huntington’s, and amyotrophic lateral sclerosis, are among the most significant health concerns worldwide, characterized by neu [...] Read more.Neha Kamboj ... Rahul KumarPublished: February 24, 2025 Explor Neurosci. 2025;4:100673
Neuroprotective insights into epigallocatechin gallate (EGCG) for neurodegenerative disordersOpen AccessReviewNeurodegenerative disorders, including Alzheimer’s, Parkinson’s, Huntington’s, and amyotrophic lateral sclerosis, are among the most significant health concerns worldwide, characterized by neu [...] Read more.Neha Kamboj ... Rahul KumarPublished: February 24, 2025 Explor Neurosci. 2025;4:100673
DOI: https://doi.org/10.37349/en.2025.100673
This article belongs to the special issue Medicinal Plants and Bioactive Phytochemicals in Neuroprotection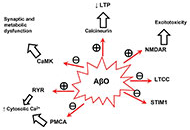 Anti-amyloid β hydrophobic peptides in Alzheimer’s disease: biomarkers and therapeutic potentialOpen AccessReviewIntracellular amyloid β oligomers (AβOs) have been linked to Alzheimer’s disease (AD) pathogenesis and to the neuronal damage in this neurodegenerative disease. Calmodulin, which binds AβO with [...] Read more.Carlos Gutierrez-MerinoPublished: February 24, 2025 Explor Neurosci. 2025;4:100672
Anti-amyloid β hydrophobic peptides in Alzheimer’s disease: biomarkers and therapeutic potentialOpen AccessReviewIntracellular amyloid β oligomers (AβOs) have been linked to Alzheimer’s disease (AD) pathogenesis and to the neuronal damage in this neurodegenerative disease. Calmodulin, which binds AβO with [...] Read more.Carlos Gutierrez-MerinoPublished: February 24, 2025 Explor Neurosci. 2025;4:100672
DOI: https://doi.org/10.37349/en.2025.100672
This article belongs to the special issue Alzheimer's Disease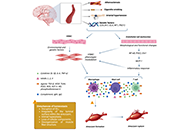 Intracranial artery dolichoectasia: What should the neurologist know?Open AccessReviewIntracranial artery dolichoectasia (IADE) is a vascular anomaly characterized by dilation and/or tortuosity of one or more intracranial arteries. While many cases are incidental findings on imaging, [...] Read more.Lorena Dellagnesi Depieri ... Lorena Souza VianaPublished: January 26, 2025 Explor Neurosci. 2025;4:100671
Intracranial artery dolichoectasia: What should the neurologist know?Open AccessReviewIntracranial artery dolichoectasia (IADE) is a vascular anomaly characterized by dilation and/or tortuosity of one or more intracranial arteries. While many cases are incidental findings on imaging, [...] Read more.Lorena Dellagnesi Depieri ... Lorena Souza VianaPublished: January 26, 2025 Explor Neurosci. 2025;4:100671
DOI: https://doi.org/10.37349/en.2025.100671
This article belongs to the special issue Cerebral Ischemia, Genetics, Comorbidities, Risk Factors and New Therapeutic Options for Neurorestoration EpilepsyOpen AccessEditorialEpilepsy, a complex and widespread neurological disorder, has evolved in its understanding from ancient misconceptions to modern scientific advancements. This special issue highlights pivotal resear [...] Read more.Jinwei ZhangPublished: January 15, 2025 Explor Neurosci. 2025;4:100670
EpilepsyOpen AccessEditorialEpilepsy, a complex and widespread neurological disorder, has evolved in its understanding from ancient misconceptions to modern scientific advancements. This special issue highlights pivotal resear [...] Read more.Jinwei ZhangPublished: January 15, 2025 Explor Neurosci. 2025;4:100670
DOI: https://doi.org/10.37349/en.2025.100670
This article belongs to the special issue Epilepsy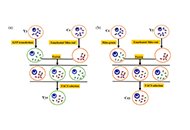 Two cellular models for analyzing mitochondrial heteroplasmyOpen AccessOriginal ArticleAim: Mitochondria are essential for brain development, and the presence of different mitochondrial types is called mitochondrial heteroplasmy. Mitochondrial dysfunction is a central aspect of man [...] Read more.Yaning Hu ... Xingbo ZhaoPublished: January 14, 2025 Explor Neurosci. 2025;4:100669
Two cellular models for analyzing mitochondrial heteroplasmyOpen AccessOriginal ArticleAim: Mitochondria are essential for brain development, and the presence of different mitochondrial types is called mitochondrial heteroplasmy. Mitochondrial dysfunction is a central aspect of man [...] Read more.Yaning Hu ... Xingbo ZhaoPublished: January 14, 2025 Explor Neurosci. 2025;4:100669
DOI: https://doi.org/10.37349/en.2025.100669
This article belongs to the special issue Mitochondria in Glia-neuron Crosstalk Protocol for measuring the benefits of exergames on executive functions under depression through a randomized multi-arm controlled experimentOpen AccessProtocolDepression is characterized by affective symptoms and neuropsychological deficits. After treatment, affective symptoms frequently remit, but cognitive alterations may remain affecting the lives of p [...] Read more.Eloisa Ruiz-MarquezPublished: December 23, 2024 Explor Neurosci. 2024;3:564–583
Protocol for measuring the benefits of exergames on executive functions under depression through a randomized multi-arm controlled experimentOpen AccessProtocolDepression is characterized by affective symptoms and neuropsychological deficits. After treatment, affective symptoms frequently remit, but cognitive alterations may remain affecting the lives of p [...] Read more.Eloisa Ruiz-MarquezPublished: December 23, 2024 Explor Neurosci. 2024;3:564–583
DOI: https://doi.org/10.37349/en.2024.00068
This article belongs to the special issue Novel Therapeutic Approaches for the Treatment of Depression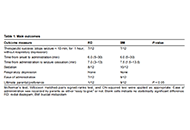 Buccal midazolam vs rectal diazepam administered by parents for continuing and serial epileptic seizures: a randomised controlled trial of parental preferencesOpen AccessOriginal ArticleAim: We aimed to undertake a small study comparing buccal midazolam and rectal diazepam in the emergency out-of-hospital treatment of prolonged or serial epileptic seizures in children. We wanted [...] Read more.Hoong Wei Gan ... William P. WhitehousePublished: December 20, 2024 Explor Neurosci. 2024;3:559–563
Buccal midazolam vs rectal diazepam administered by parents for continuing and serial epileptic seizures: a randomised controlled trial of parental preferencesOpen AccessOriginal ArticleAim: We aimed to undertake a small study comparing buccal midazolam and rectal diazepam in the emergency out-of-hospital treatment of prolonged or serial epileptic seizures in children. We wanted [...] Read more.Hoong Wei Gan ... William P. WhitehousePublished: December 20, 2024 Explor Neurosci. 2024;3:559–563
DOI: https://doi.org/10.37349/en.2024.00067
This article belongs to the special issue Advances in Epilepsy Research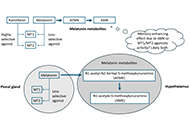 What might melatonin supplementation provide for humans beyond improved onset to sleep?Open AccessPerspectiveMelatonin is widely available as a dietary supplement and/or medicine for sleep. It is an endogenous hormone produced in the pineal gland of the brain, with metabolites providing additional benefici [...] Read more.Leticia A. SheaPublished: November 26, 2024 Explor Neurosci. 2024;3:551–558
What might melatonin supplementation provide for humans beyond improved onset to sleep?Open AccessPerspectiveMelatonin is widely available as a dietary supplement and/or medicine for sleep. It is an endogenous hormone produced in the pineal gland of the brain, with metabolites providing additional benefici [...] Read more.Leticia A. SheaPublished: November 26, 2024 Explor Neurosci. 2024;3:551–558
DOI: https://doi.org/10.37349/en.2024.00066
This article belongs to the special issue Circadian Rhythm and Melatonin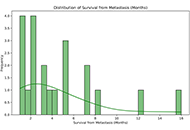 Metastatic glioblastoma multiforme on skin and subcutaneous tissueOpen AccessReviewGlioblastoma multiforme (GBM) is characterized by its infiltrative growth pattern and high recurrence rate despite treatment. While local progression within the central nervous system (CNS) is the r [...] Read more.Maria Ciscar-Fabuel ... Andreu Gabarros-CanalsPublished: October 29, 2024 Explor Neurosci. 2024;3:539–550
Metastatic glioblastoma multiforme on skin and subcutaneous tissueOpen AccessReviewGlioblastoma multiforme (GBM) is characterized by its infiltrative growth pattern and high recurrence rate despite treatment. While local progression within the central nervous system (CNS) is the r [...] Read more.Maria Ciscar-Fabuel ... Andreu Gabarros-CanalsPublished: October 29, 2024 Explor Neurosci. 2024;3:539–550
DOI: https://doi.org/10.37349/en.2024.00065
This article belongs to the special issue Current Approaches to Malignant Tumors of the Nervous System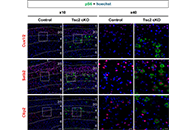 Somatostatin-expressing inhibitory neurons with mTORC1 activation in cortical layers 4/5 are involved in the epileptogenesis of miceOpen AccessOriginal ArticleAim: Patients with tuberous sclerosis complex (TSC) which is caused by hyperactivation of mechanistic target of rapamycin complex 1 (mTORC1) often show giant cells in the brain. These giant cells [...] Read more.Fumiki Yamashita ... Mari Wataya-KanedaPublished: October 29, 2024 Explor Neurosci. 2024;3:527–538
Somatostatin-expressing inhibitory neurons with mTORC1 activation in cortical layers 4/5 are involved in the epileptogenesis of miceOpen AccessOriginal ArticleAim: Patients with tuberous sclerosis complex (TSC) which is caused by hyperactivation of mechanistic target of rapamycin complex 1 (mTORC1) often show giant cells in the brain. These giant cells [...] Read more.Fumiki Yamashita ... Mari Wataya-KanedaPublished: October 29, 2024 Explor Neurosci. 2024;3:527–538
DOI: https://doi.org/10.37349/en.2024.00064
This article belongs to the special issue Epilepsy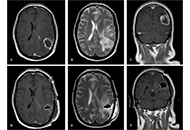 Glioblastoma multiforme recurrence on skin and subcutaneous tissue: a case reportOpen AccessCase ReportGlioblastoma multiforme (GBM) is the most common malignant primary central nervous system (CNS) tumor. It presents an aggressive pattern, with a tendency for intracranial progression despite optimal [...] Read more.Maria Ciscar-Fabuel ... Andreu Gabarros-CanalsPublished: October 16, 2024 Explor Neurosci. 2024;3:520–526
Glioblastoma multiforme recurrence on skin and subcutaneous tissue: a case reportOpen AccessCase ReportGlioblastoma multiforme (GBM) is the most common malignant primary central nervous system (CNS) tumor. It presents an aggressive pattern, with a tendency for intracranial progression despite optimal [...] Read more.Maria Ciscar-Fabuel ... Andreu Gabarros-CanalsPublished: October 16, 2024 Explor Neurosci. 2024;3:520–526
DOI: https://doi.org/10.37349/en.2024.00063
This article belongs to the special issue Current Approaches to Malignant Tumors of the Nervous System Epilepsy adverse events post vaccinationOpen AccessOriginal ArticleAim: Seizure and epilepsy adverse events (AEs) can occur following vaccination. For epilepsy AEs, they are generally expected to only occur at background population frequencies without associatio [...] Read more.Darrell O. RickePublished: October 16, 2024 Explor Neurosci. 2024;3:508–519
Epilepsy adverse events post vaccinationOpen AccessOriginal ArticleAim: Seizure and epilepsy adverse events (AEs) can occur following vaccination. For epilepsy AEs, they are generally expected to only occur at background population frequencies without associatio [...] Read more.Darrell O. RickePublished: October 16, 2024 Explor Neurosci. 2024;3:508–519
DOI: https://doi.org/10.37349/en.2024.00062
This article belongs to the special issue Epilepsy Observing nociceptive detection thresholds and evoked potentials in diabetic patients with and without painful neuropathyOpen AccessOriginal ArticleAim: Diabetic polyneuropathy is the most described complication in patients with diabetes mellitus. A significant percentage of these patients experience disabling neuropathic pain (painful diabe [...] Read more.Tom Berfelo ... Jan R. BuitenwegPublished: October 15, 2024 Explor Neurosci. 2024;3:493–507
Observing nociceptive detection thresholds and evoked potentials in diabetic patients with and without painful neuropathyOpen AccessOriginal ArticleAim: Diabetic polyneuropathy is the most described complication in patients with diabetes mellitus. A significant percentage of these patients experience disabling neuropathic pain (painful diabe [...] Read more.Tom Berfelo ... Jan R. BuitenwegPublished: October 15, 2024 Explor Neurosci. 2024;3:493–507
DOI: https://doi.org/10.37349/en.2024.00061
This article belongs to the special issue Neuropathic Pain A review of epileptic markers: from ion channels, astrocytes, synaptic imbalance to whole brain network dynamicsOpen AccessReviewEpilepsy, a neurological disorder characterized by recurrent seizures, presents a complex interplay of cellular and molecular mechanisms. The symptoms manifest themselves at various scales, from ion [...] Read more.Swati Banerjee, Viktor JirsaPublished: September 29, 2024 Explor Neurosci. 2024;3:478–492
A review of epileptic markers: from ion channels, astrocytes, synaptic imbalance to whole brain network dynamicsOpen AccessReviewEpilepsy, a neurological disorder characterized by recurrent seizures, presents a complex interplay of cellular and molecular mechanisms. The symptoms manifest themselves at various scales, from ion [...] Read more.Swati Banerjee, Viktor JirsaPublished: September 29, 2024 Explor Neurosci. 2024;3:478–492
DOI: https://doi.org/10.37349/en.2024.00060
This article belongs to the special issue Epilepsy -
